To quantify the Spanish Pharmacy and Therapeutics Commission (P&TC) activity with regard to assessing and selecting drugs and describing variability in decisions made to include them.
MethodDescriptive, cross-sectional study based on a questionnaire aimed at 513 hospitals with more than 75 beds. We included questions referring to the P&TC resolutions, the therapeutic positioning and assessment reports. Recruitment was carried out between November 2007 and January 2008. Variability among P&TC conclusions was presented in five categories or levels of coincidence.
ResultsOne hundred and seventy-five hospitals participated, with a response rate of 34% (54% of beds). The mean number of drug-indications assessed per hospital was 10.35 (7.45). The proportion of assessments that conclude with drug inclusion or rejection was 75.3% and 21.4%, respectively. 16.2% concluded with therapeutic equivalence. Conditions for use were established for 64% of them, and 33% were included in a clinical guide. With regard to variability, 81.0% of assessments coincided with the conclusion to include or reject the drug. A contradictory decision was made for 19.0%.
ConclusionsDrug assessment and selection in hospitals are considerable. The proportion of drugs approved is similar in different types of hospitals. There is extensive variability as regards deciding upon inclusion and is similar to studies conducted in other countries. They indicate that a standardising methodology would be recommendable.
Cuantificar la actividad de las Comisiones de Farmacia y Terapéutica (CFyT) con relación a la evaluación y selección de medicamentos, y describir la variabilidad en las decisiones de incorporación de los mismos.
MétodoEstudio descriptivo transversal basado en un cuestionario dirigido a los 513 hospitales españoles con más de 75 camas. Se incluyeron preguntas referidas a las resoluciones de la CFyT, el posicionamiento terapéutico y los informes de evaluación. La selección se realizó entre noviembre de 2007 y enero de 2008. La variabilidad en las conclusiones de las CFyT se expresa en 5 categorías o grados de coincidencia.
ResultadosParticiparon 175 hospitales, tasa de respuesta del 34% (54% de las camas). El número medio de medicamentos-indicación evaluados por hospital fue 10,35 (7,45). La proporción de evaluaciones que concluyen en inclusión o rechazo del fármaco fue del 75,3 y 21,4%, respectivamente. En el 16,2% se concluyó en equivalencia terapéutica. Se establecieron condiciones de uso en un 64%, y se incluyeron en un guía clínica en un 33%. En cuanto a la variabilidad, en el 81,0% de las evaluaciones realizadas se coincide en la conclusión de incluir o de rechazar el medicamento, en el 19,0% se ha tomado la decisión opuesta a la mayoritaria.
ConclusionesLa actividad de evaluación y selección de medicamentos en los hospitales es considerable. La proporción de medicamentos aprobados es similar en los diferentes tipos de hospital. La variabilidad en la decisión de inclusión es amplia y similar a estudios realizados en otros países. Indican la conveniencia de estandarización de la metodología.
Hospitals have different means of establishing policies oriented towards promoting safe, effective and efficient medication use. Assessing and selecting drugs and making decisions about what to include in drug formularies (DF) are undertaken by Pharmacy and Therapeutics Committees (P&TCs).
Until just a short while ago, there were no available data on the structural and functional organisation of P&TCs in Spanish hospitals. However, the first results from a study carried out in 2007 and 2008 with the participation of 200 hospitals were published recently. This study describes the structure and function of P&TCs in Spain, which are comparable to those in other developed countries. It also describes assessment and selection procedures, which are very similar among Spanish hospitals of different types and sizes.1
Likewise, no detailed information was available regarding P&TC activities, such as information about the drugs these committees evaluated and their decisions as to what to incorporate in DFs. Spain is currently witnessing the development of the project Atlas de variaciones en la práctica médica en el Sistema Nacional de Salud, Red de Investigación en Resultados y Servicios de Salud (VPM-IRYSS) (Atlas of medical practice variations in the National Health System, carried out by the healthcare results and services research network).2,3 Its purpose is to systematically describe variations in the use of health services pertaining to Spain's National Health System. With this in mind, gaining an understanding of the variability present in the drug selection and decision-making processes among different P&TCs is an interesting prospect, as this variability certainly affects the lists of drugs available at each hospital.
The purpose of this study is to quantify how P&TCs in Spanish hospitals undertake drug assessment and selection processes, and to describe different decision-making approaches used by hospitals for including drugs.
MethodDescriptive, cross-sectional study based on a questionnaire sent to the 541 Spanish hospitals with more than 75 beds, according to the Spanish catalogue of hospitals.4 From this list we excluded hospital morgues and hospitals that form part of another centre (n=28). The final study population included 513 hospitals.
The questionnaire, addressed to pharmacy service directors and P&TC secretaries, inquired about drugs studied by the P&TC during 2006. The questionnaire included a list of drugs that were authorised in Spain between 2004 and 2006 (generic and trade names and clinical indication), leaving additional space for information about drugs that became commercially available before that time. The questionnaire included 7 questions per drug and indication. The first 2 referred to P&TC decisions regarding including drugs and declaring them as therapeutic equivalents; the next 2 referred to establishing conditions of use and incorporating them into formularies or clinical protocols. The final 3 questions addressed whether or not a drug evaluation report was available, report conclusions where applicable, and whether or not they coincided with the P&TC's decision. Each question had 3 possible answers (yes, no, don’t know); the question regarding drug inclusion had a fourth answer (decision postponed). The questionnaire's final wording was based on a pilot study in which 16 hospitals similar to those in our study population participated.
The questionnaire was designed as an online tool and included drop-down menus and check boxes. It was found on the webpage of an online platform. Recruitment took place between November 2007 and January 2008 by postal mail (2 mailings), e-mail (3 mailings) and telephone communications (2 calls). Throughout the entire process, including processing the results, we received technical support from the Unidad de Apoyo Metodológico a la Investigación de la Escuela Andaluza de Salud Pública (EASP) (Research methodology support unit at the Andalusian school of public health).
Descriptive data analysis was performed using frequency and percentage tables for qualitative variables and numerical summaries including the minimum and maximum values, mean, and standard deviation for quantitative variables. Bivariate analysis was used to analyse the association between questionnaire variables, the size of the hospital, and its status as public or private and teaching or non-teaching. Hospitals were classified by size as follows: less than 100 beds, 100–199 beds, 200–499 beds, and 500 beds or more. We analysed associations for drugs dispensed by prescription and drugs for hospital use or diagnostic purposes. The analysis made use of the chi-squared test, Fisher's exact test, Student's t-test and the Kruskal–Wallis H-test as needed.
The variability of conclusions reached by P&TCs was obtained based on drug-indications assessed by more than 2 hospitals. Variability was expressed as 5 categories or degrees of agreement: Total agreement (100% of the drug assessments coincided to approve the drug); high agreement (between 85.0% and 99.9% of the assessments approved the drug); moderate agreement (between 70.0% and 84.9% of the assessments coincided); low agreement (between 50.0% and 69.9% of the assessments coincided) and very low agreement (less than 50% of the assessments coincided, meaning that these drugs were generally denied approval).
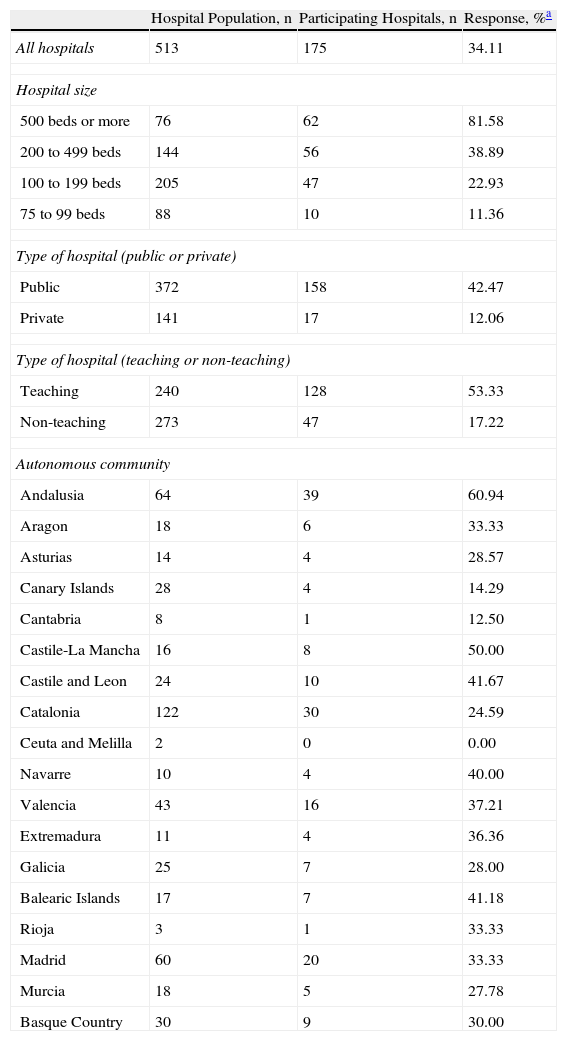
ResultsResponseA total of 175 hospitals completed the questionnaire (response rate of 34.11%); together, they represented 54% of the total number of beds in the hospitals that received the survey. Hospitals from all of Spain's autonomous communities, except for Ceuta and Melilla, participated in the survey. Larger hospitals, public hospitals and teaching hospitals had higher participation rates than the rest (Table 1).
Characteristics of the Study Population, Participating Hospitals and Response Rates.
| Hospital Population, n | Participating Hospitals, n | Response, %a | |
| All hospitals | 513 | 175 | 34.11 |
| Hospital size | |||
| 500 beds or more | 76 | 62 | 81.58 |
| 200 to 499 beds | 144 | 56 | 38.89 |
| 100 to 199 beds | 205 | 47 | 22.93 |
| 75 to 99 beds | 88 | 10 | 11.36 |
| Type of hospital (public or private) | |||
| Public | 372 | 158 | 42.47 |
| Private | 141 | 17 | 12.06 |
| Type of hospital (teaching or non-teaching) | |||
| Teaching | 240 | 128 | 53.33 |
| Non-teaching | 273 | 47 | 17.22 |
| Autonomous community | |||
| Andalusia | 64 | 39 | 60.94 |
| Aragon | 18 | 6 | 33.33 |
| Asturias | 14 | 4 | 28.57 |
| Canary Islands | 28 | 4 | 14.29 |
| Cantabria | 8 | 1 | 12.50 |
| Castile-La Mancha | 16 | 8 | 50.00 |
| Castile and Leon | 24 | 10 | 41.67 |
| Catalonia | 122 | 30 | 24.59 |
| Ceuta and Melilla | 2 | 0 | 0.00 |
| Navarre | 10 | 4 | 40.00 |
| Valencia | 43 | 16 | 37.21 |
| Extremadura | 11 | 4 | 36.36 |
| Galicia | 25 | 7 | 28.00 |
| Balearic Islands | 17 | 7 | 41.18 |
| Rioja | 3 | 1 | 33.33 |
| Madrid | 60 | 20 | 33.33 |
| Murcia | 18 | 5 | 27.78 |
| Basque Country | 30 | 9 | 30.00 |
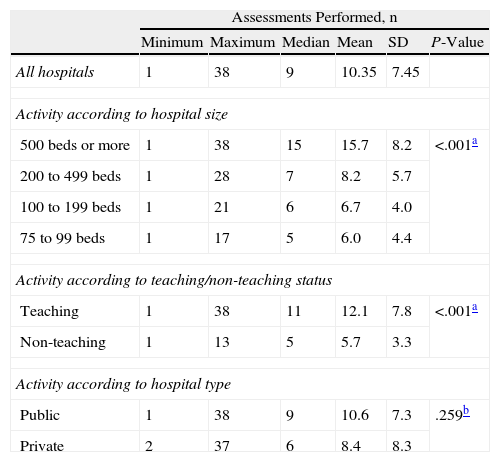
A total of 356 different drug-indications were evaluated in 2006, and total completed assessments came to 1805. The mean number (SD) of drugs evaluated per hospital was 10.35 (7.45). Drugs evaluated by more than 1 hospital totalled 171; 71 of these were new drugs and indications authorised in Spain during 2004–2006, and 100 were authorised prior to 2004.
Larger-capacity hospitals completed more assessments than smaller ones, and teaching hospitals completed more assessments than non-teaching ones. There were no differences between public and private hospitals (Table 2).
Drug-Indications Evaluated by the Pharmacy and Therapeutics Committee in 2006. Overall Activity Data, According to Hospital Size, Teaching or Non-Teaching Status and Hospital Type.
| Assessments Performed, n | ||||||
| Minimum | Maximum | Median | Mean | SD | P-Value | |
| All hospitals | 1 | 38 | 9 | 10.35 | 7.45 | |
| Activity according to hospital size | ||||||
| 500 beds or more | 1 | 38 | 15 | 15.7 | 8.2 | <.001a |
| 200 to 499 beds | 1 | 28 | 7 | 8.2 | 5.7 | |
| 100 to 199 beds | 1 | 21 | 6 | 6.7 | 4.0 | |
| 75 to 99 beds | 1 | 17 | 5 | 6.0 | 4.4 | |
| Activity according to teaching/non-teaching status | ||||||
| Teaching | 1 | 38 | 11 | 12.1 | 7.8 | <.001a |
| Non-teaching | 1 | 13 | 5 | 5.7 | 3.3 | |
| Activity according to hospital type | ||||||
| Public | 1 | 38 | 9 | 10.6 | 7.3 | .259b |
| Private | 2 | 37 | 6 | 8.4 | 8.3 | |
SD: standard deviation.
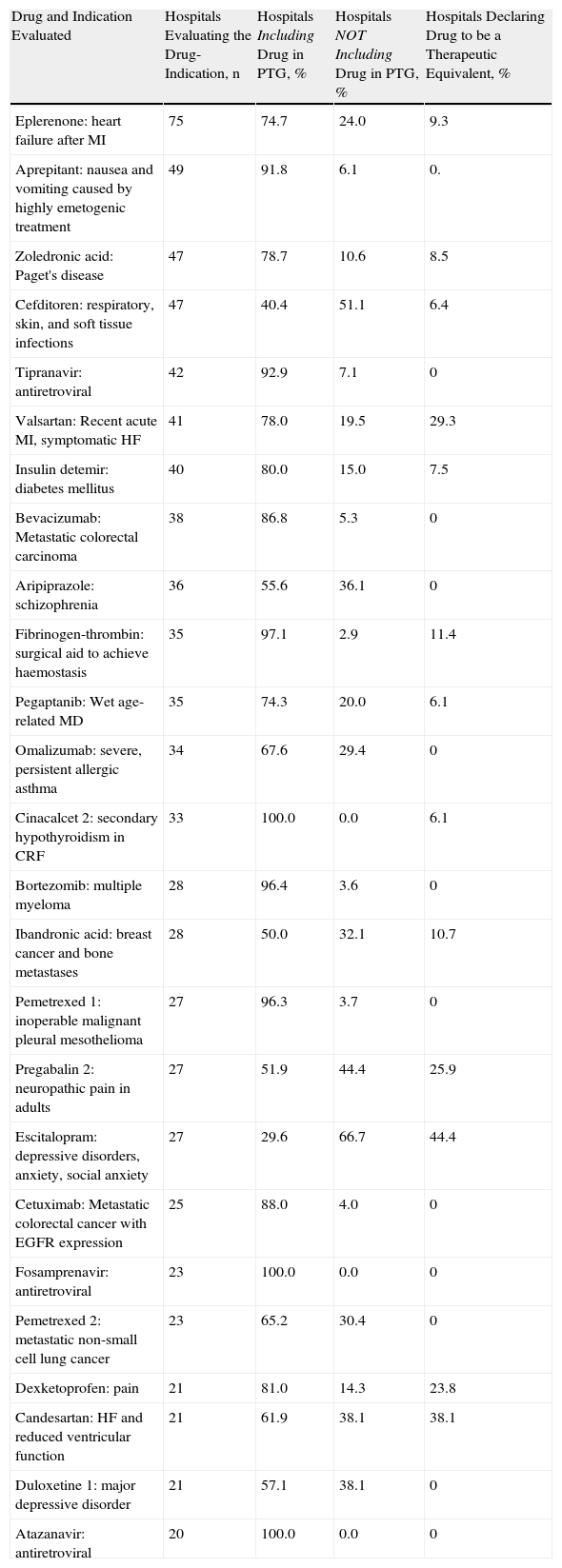
Assessments were completed by 20 or more hospitals in the case of 25 drug-indications; by between 10 and 20 hospitals for 22 drugs; and between 3 and 10 hospitals for 82 drugs. The most commonly evaluated drugs were eplerenone, aprepitant, zoledronic acid and cefditoren (Table 3).
Drugs and Indications Most Commonly Evaluated in Hospitals in 2006. Number and Percentage of Hospitals Resolving to Approve or Refuse a Drug or Declare it a Therapeutic Equivalenta (List of Drugs Evaluated by 20 Hospitals or More).
| Drug and Indication Evaluated | Hospitals Evaluating the Drug-Indication, n | Hospitals Including Drug in PTG, % | Hospitals NOT Including Drug in PTG, % | Hospitals Declaring Drug to be a Therapeutic Equivalent, % |
| Eplerenone: heart failure after MI | 75 | 74.7 | 24.0 | 9.3 |
| Aprepitant: nausea and vomiting caused by highly emetogenic treatment | 49 | 91.8 | 6.1 | 0. |
| Zoledronic acid: Paget's disease | 47 | 78.7 | 10.6 | 8.5 |
| Cefditoren: respiratory, skin, and soft tissue infections | 47 | 40.4 | 51.1 | 6.4 |
| Tipranavir: antiretroviral | 42 | 92.9 | 7.1 | 0 |
| Valsartan: Recent acute MI, symptomatic HF | 41 | 78.0 | 19.5 | 29.3 |
| Insulin detemir: diabetes mellitus | 40 | 80.0 | 15.0 | 7.5 |
| Bevacizumab: Metastatic colorectal carcinoma | 38 | 86.8 | 5.3 | 0 |
| Aripiprazole: schizophrenia | 36 | 55.6 | 36.1 | 0 |
| Fibrinogen-thrombin: surgical aid to achieve haemostasis | 35 | 97.1 | 2.9 | 11.4 |
| Pegaptanib: Wet age-related MD | 35 | 74.3 | 20.0 | 6.1 |
| Omalizumab: severe, persistent allergic asthma | 34 | 67.6 | 29.4 | 0 |
| Cinacalcet 2: secondary hypothyroidism in CRF | 33 | 100.0 | 0.0 | 6.1 |
| Bortezomib: multiple myeloma | 28 | 96.4 | 3.6 | 0 |
| Ibandronic acid: breast cancer and bone metastases | 28 | 50.0 | 32.1 | 10.7 |
| Pemetrexed 1: inoperable malignant pleural mesothelioma | 27 | 96.3 | 3.7 | 0 |
| Pregabalin 2: neuropathic pain in adults | 27 | 51.9 | 44.4 | 25.9 |
| Escitalopram: depressive disorders, anxiety, social anxiety | 27 | 29.6 | 66.7 | 44.4 |
| Cetuximab: Metastatic colorectal cancer with EGFR expression | 25 | 88.0 | 4.0 | 0 |
| Fosamprenavir: antiretroviral | 23 | 100.0 | 0.0 | 0 |
| Pemetrexed 2: metastatic non-small cell lung cancer | 23 | 65.2 | 30.4 | 0 |
| Dexketoprofen: pain | 21 | 81.0 | 14.3 | 23.8 |
| Candesartan: HF and reduced ventricular function | 21 | 61.9 | 38.1 | 38.1 |
| Duloxetine 1: major depressive disorder | 21 | 57.1 | 38.1 | 0 |
| Atazanavir: antiretroviral | 20 | 100.0 | 0.0 | 0 |
PTG: Pharmacotherapeutic guidelines.
The discrepancy between the percentages of hospitals either approving or refusing the drug and the total is explained by the percentage of hospitals that postponed the decision (this value is not shown in the table). The percentage of therapeutic equivalents corresponds to a survey question that was independent from the drug's approval/refusal status (a therapeutic equivalent could be declared even if the drug was refused).
The mean number (SD) of completed hospital assessments which resulted in inclusion of the drug was 7.79 (5.69); the mean number of those denying approval was 2.22 (2.73) and those postponing the decision, 0.33 (0.77). These figures represent 75.3%, 21.4%, and 3.2%, respectively, of the total completed assessments. The percentage of the assessments resulting in inclusion does not differ according to whether or not the hospital is a teaching hospital or public or private. We observed no differences among hospitals with capacities greater than or less than 500 beds.
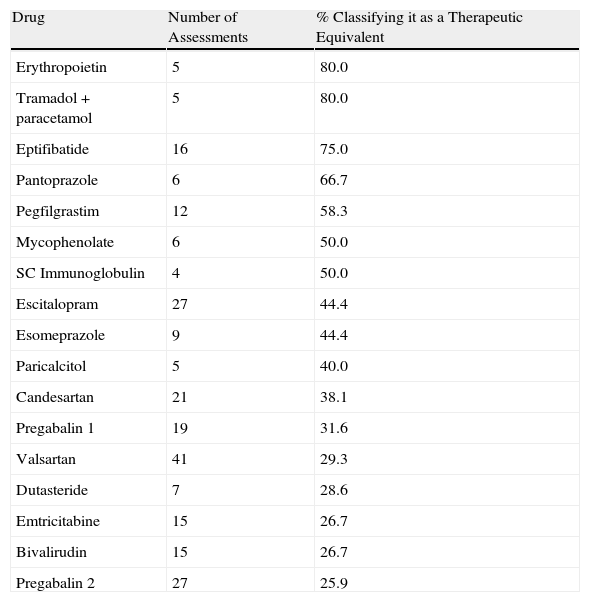
At total of 62 drugs were declared therapeutic equivalents by at least 1 assessment. The mean number (SD) of completed assessments per hospital resulting in a drug being declared a therapeutic equivalent was 1.26 (1.95), which represents 16.2% of the assessment total. Hospitals with a capacity of more than 500 beds have a higher mean (SD) number of drugs declared therapeutic equivalents than smaller centres do: 1.98 (2.65) vs 0.87 (1.27) (P=.005). However, the number of drugs declared equivalents as a fraction of the total evaluated by each hospital does not present significant differences among hospitals of different sizes. The percentages of favourable assessments for the most commonly evaluated drugs are shown in Table 3 and the list of drugs considered to be equivalents by at least 25% of the evaluating hospitals are shown in Table 4.
List of Drugs Considered to be Therapeutic Equivalents by at Least 25% of the Surveyed Hospitals.
| Drug | Number of Assessments | % Classifying it as a Therapeutic Equivalent |
| Erythropoietin | 5 | 80.0 |
| Tramadol+paracetamol | 5 | 80.0 |
| Eptifibatide | 16 | 75.0 |
| Pantoprazole | 6 | 66.7 |
| Pegfilgrastim | 12 | 58.3 |
| Mycophenolate | 6 | 50.0 |
| SC Immunoglobulin | 4 | 50.0 |
| Escitalopram | 27 | 44.4 |
| Esomeprazole | 9 | 44.4 |
| Paricalcitol | 5 | 40.0 |
| Candesartan | 21 | 38.1 |
| Pregabalin 1 | 19 | 31.6 |
| Valsartan | 41 | 29.3 |
| Dutasteride | 7 | 28.6 |
| Emtricitabine | 15 | 26.7 |
| Bivalirudin | 15 | 26.7 |
| Pregabalin 2 | 27 | 25.9 |
Conditions of use were established for a mean (SD) number of 4.99 (5.02) drugs; a mean of 2.57 (3.50) was included in a clinical protocol or clinical guidelines. Respectively, those figures represent 64% and 33% of the assessments proposing inclusion. Centres with more than 500 beds established conditions of use more often than smaller hospitals did, in 70.7% and 56.3% of the cases respectively (P<.001). Drug incorporation in clinical guidelines and protocols was 35.6% in hospitals with a capacity of 500 beds or more, and 29.9% in smaller hospitals (P=.025).
With regard to dispensing method, diagnostic and hospital-use drugs were more commonly included than drugs dispensed by prescription, at 82.5% and 54.2% respectively (P<.001). On the other hand, we observe a higher percentage of drugs declared therapeutic equivalents among drugs dispensed by prescription than among those for diagnostic and hospital use: 26.1% vs 11.6% (P<.001).
An evaluation report is available for a mean number (SD) of 7.95 (7.00) assessments, representing 77% of the completed assessments. The conclusion reached by the P&TC was the same as that in the report on a mean number of 7.07 (6.78) occasions, or 89%.
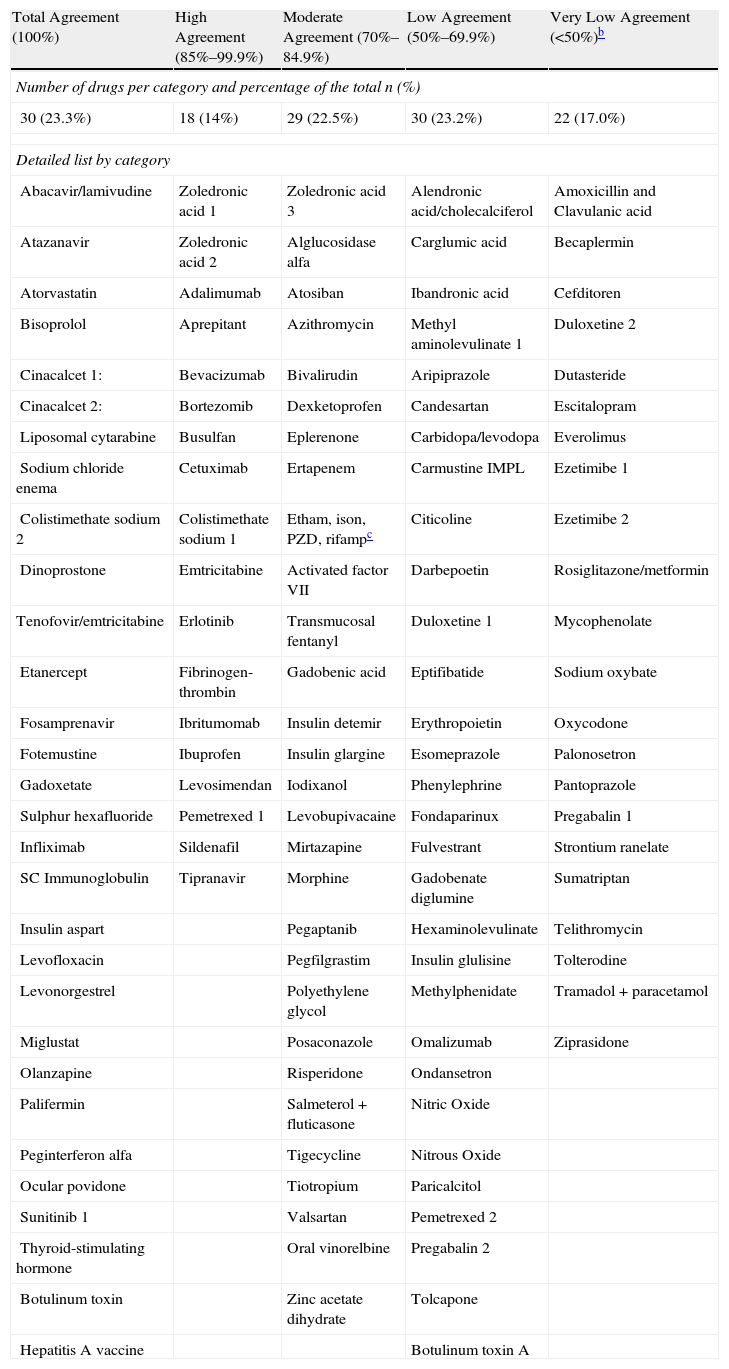
Variability in SelectionOf the total drug-indications studied, the 129 drugs that were assessed by more than 2 hospitals, account for 1558 assessments. The decision to incorporate the drug was postponed in 53 assessments, and our study is therefore based on 1505 assessments. Variability in selection results is expressed by the degree of agreement in the decision (Table 5).
Degree of Agreement in the Decision to Incorporate the Drug. Classified by Categories.a
| Total Agreement (100%) | High Agreement (85%–99.9%) | Moderate Agreement (70%–84.9%) | Low Agreement (50%–69.9%) | Very Low Agreement (<50%)b |
| Number of drugs per category and percentage of the total n (%) | ||||
| 30 (23.3%) | 18 (14%) | 29 (22.5%) | 30 (23.2%) | 22 (17.0%) |
| Detailed list by category | ||||
| Abacavir/lamivudine | Zoledronic acid 1 | Zoledronic acid 3 | Alendronic acid/cholecalciferol | Amoxicillin and Clavulanic acid |
| Atazanavir | Zoledronic acid 2 | Alglucosidase alfa | Carglumic acid | Becaplermin |
| Atorvastatin | Adalimumab | Atosiban | Ibandronic acid | Cefditoren |
| Bisoprolol | Aprepitant | Azithromycin | Methyl aminolevulinate 1 | Duloxetine 2 |
| Cinacalcet 1: | Bevacizumab | Bivalirudin | Aripiprazole | Dutasteride |
| Cinacalcet 2: | Bortezomib | Dexketoprofen | Candesartan | Escitalopram |
| Liposomal cytarabine | Busulfan | Eplerenone | Carbidopa/levodopa | Everolimus |
| Sodium chloride enema | Cetuximab | Ertapenem | Carmustine IMPL | Ezetimibe 1 |
| Colistimethate sodium 2 | Colistimethate sodium 1 | Etham, ison, PZD, rifampc | Citicoline | Ezetimibe 2 |
| Dinoprostone | Emtricitabine | Activated factor VII | Darbepoetin | Rosiglitazone/metformin |
| Tenofovir/emtricitabine | Erlotinib | Transmucosal fentanyl | Duloxetine 1 | Mycophenolate |
| Etanercept | Fibrinogen-thrombin | Gadobenic acid | Eptifibatide | Sodium oxybate |
| Fosamprenavir | Ibritumomab | Insulin detemir | Erythropoietin | Oxycodone |
| Fotemustine | Ibuprofen | Insulin glargine | Esomeprazole | Palonosetron |
| Gadoxetate | Levosimendan | Iodixanol | Phenylephrine | Pantoprazole |
| Sulphur hexafluoride | Pemetrexed 1 | Levobupivacaine | Fondaparinux | Pregabalin 1 |
| Infliximab | Sildenafil | Mirtazapine | Fulvestrant | Strontium ranelate |
| SC Immunoglobulin | Tipranavir | Morphine | Gadobenate diglumine | Sumatriptan |
| Insulin aspart | Pegaptanib | Hexaminolevulinate | Telithromycin | |
| Levofloxacin | Pegfilgrastim | Insulin glulisine | Tolterodine | |
| Levonorgestrel | Polyethylene glycol | Methylphenidate | Tramadol+paracetamol | |
| Miglustat | Posaconazole | Omalizumab | Ziprasidone | |
| Olanzapine | Risperidone | Ondansetron | ||
| Palifermin | Salmeterol+fluticasone | Nitric Oxide | ||
| Peginterferon alfa | Tigecycline | Nitrous Oxide | ||
| Ocular povidone | Tiotropium | Paricalcitol | ||
| Sunitinib 1 | Valsartan | Pemetrexed 2 | ||
| Thyroid-stimulating hormone | Oral vinorelbine | Pregabalin 2 | ||
| Botulinum toxin | Zinc acetate dihydrate | Tolcapone | ||
| Hepatitis A vaccine | Botulinum toxin A | |||
In general, 81.0% of the assessments coincided with others in the decision to include or to refuse the drug; in the remaining 19.0%, the decision opposed that of the majority.
We found 32 drugs that at least 2 hospitals considered to be therapeutic equivalents. Only 5 drugs were classified as therapeutic equivalents by more than 50% of the hospitals (erythropoietin, eptifibatide, pantoprazole, pegfilgrastim and tramadol-paracetamol). A total of 16 drugs were considered equivalents by at least 25% of the evaluating hospitals (Table 4).
Overall, in 21.3% of the 686 assessments completed for drugs declared to be equivalents, the hospitals supported the declaration. In the remaining 78.7% of the assessments, they did not come to the same conclusion.
DiscussionThis study evaluates activity carried out by P&TCs in Spain. Overall hospital participation in the study exceeded one third of the sample size and was representative in that it included hospitals from 15 of 17 autonomous communities, containing more than half of all Spanish hospital beds. The higher percentage of participation among large, public and teaching hospitals may constitute a bias, since these are the hospitals which also engage in the most drug assessment and selection activity.
The mean yearly number of drugs evaluated by Spanish hospitals shows that P&TCs are highly active, although it presents a wide range which shows that this activity is distributed unevenly between different hospitals. A total of 356 different drug-indications were evaluated. Approximately half were evaluated only once, and most were drugs that have been commercially available for years. Assessment was therefore undertaken in response to specific needs for drug incorporation that had not been evaluated previously.
More drug assessment activity takes place in larger, teaching hospitals which are also the ones with higher capacities. No differences were found between public and private centres. These variations are logical because larger, teaching hospitals tend to be associated with medical complexity.
The mean yearly number of assessments performed per hospital (approximately 10) is significantly lower than the number of new active ingredients entering the market or the number of new indications approved in Spain (annual means are 21 and 40, respectively).5 This could be due to the fact that inclusion request circuits for new drugs and the assessment process itself both limit the number of evaluated drugs, so that assessments are directed mainly towards drugs that may potentially provide therapeutic benefits.
The list of most commonly evaluated drugs in 2006 contains a high percentage of drugs indicated for patients receiving care and follow-up in hospitals, such as drugs used in treating cancer or HIV. Evaluating these types of drugs is practically mandatory for hospitals that treat such patients. However, drugs mainly used in an outpatient setting were also evaluated. These include medications which documentation centres belonging to the Spanish joint committee for new drug assessment (Comité Mixto de Evaluación de Nuevos Medicamentos, CMENM)6 have classified as having therapeutic use (atorvastatin, eplerenone, valsartan) and others, classified by that committee as “drugs providing no new benefits”, whose added therapeutic value is disputed (aripiprazole, cefditoren, duloxetine, escitalopram, esomeprazole, pregabalin, and others). It is possible that demand for hospitals to incorporate some of the latter was created through advertising.
Numerous drugs were studied by different hospitals during the same year. A more efficient strategy would place emphasis on inter-hospital cooperation so as not to duplicate efforts unnecessarily. In fact, such cooperation is already partly underway, through the exchange of information that takes place in the SEFH's GENESIS group.7,8 In response to our survey question regarding use of the reports published on the GENESIS webpage, 85.5% of the hospitals stated that they consult the reports and consider them to be useful sources of information, and 44.5% stated that they are always or usually helpful to the hospital's P&TC (data not shown).
The conclusion of 3 out of 4 hospital assessments is approval of the drug. This statistic is in keeping with the fact, mentioned previously that only a fraction of the drugs commercialised in Spain receive a request for hospital assessment, and that these are likely to be drugs with a higher probability of being approved. This percentage is somewhat higher than those published by some specific hospitals whose drug approval percentages range between 62.5%9 and 68%.10
On the other hand, while larger hospitals and teaching hospitals engage in more assessment activities, the percentage of drugs approved and refused is similar for all hospitals. This seems to indicate that evaluation and decision-making criteria are similar for different hospital types and sizes, and reflects the fact that the selection procedures and systems used by different kinds of Spanish hospitals share essential aspects.1
Often, conditions of use are set forth, or the drugs are incorporated into treatment protocols. This indicates that P&TC activity is not limited to issuing dichotomous rulings on whether or not to incorporate drugs into the hospital formulary; it goes so far as to place the drug within a treatment regimen.
The share of assessments defining 2 or more drugs as therapeutic equivalents is sizeable, approximately 1 in 6. This is a very important criterion in Spanish hospitals, as drug equivalency is taken to mean absence of added therapeutic value, allowing us to apply efficiency criteria when managing purchases.11
In our study, conclusions coincide in approximately 4 out of 5 completed assessments; hospital decisions run counter to the majority in 1 of 5 assessments, which indicates a small, yet significant, degree of variability. We should mention that in just over one third of the drug studies, all or nearly all of the hospitals coincided in including the drug. Most of these drugs are for hospital use. On the other hand, 17.1% of the drugs were refused by a majority of hospitals; most of these are dispensed by prescription and are listed in the “drugs providing no new benefits” category according to assessments published by the CMENM.6 Lastly, for nearly half of the drugs, the degree of agreement is moderate to low. These are the drugs for which there is the highest variability among decisions to include them or not.
Published studies employ a number of different methods to express variability in the selection process, or else simply present the study result in the form of percentages. This is because Cohen's kappa, the coefficient of reference for evaluating variability in other areas, does not properly interpret the degree of disagreement for the content of a questionnaire.12
In a survey of 41 healthcare facilities in the United States, Dranove et al.13 studied the incorporation of 7 drugs in drug formularies and found variability of incorporation ranging from 25% to 80%. Shrank et al.14 studied variability in 5 drug groups in 6 health plans provided by insurance companies in California; in the year 2002 no single drug was found in all of the formularies, 10% were in just 1, 30% were in 5 and 60% were found in between 2 and 4 formularies. Tseng et al.15 studied 72 Medicare formularies and evaluated the presence of 75 drugs. They found that mean coverage of drug formularies was 69% with a range of 7%–100%.
In a study focusing on 6 new cardiovascular drugs and their assessments by 164 P&TCs in Canadian hospitals, Shalansky et al.16 observed that the percentage of inclusion was 81% for abciximab, 33% for enoxaparin, 20% for dalteparin, 43% for clopidogrel, 12% for eptifibatide and 9% for tirofiban. In another Canadian study of a sample of 58 drugs approved between 1996 and 1997, only 9% were to be found on all of the formularies corresponding to 10 provinces; 24% were found on at least 8, and the level of agreement measured by Cohen's kappa was 0.20.17 Other Canadian studies also show low agreement indices.18,19 More recently, Morgan et al.12 analysed drug formularies from 9 Canadian provinces and found a range of 55%–76% incorporation for the 765 available drugs. Only 41% of the drugs were found on all of the formularies.
In a study of 143 participating German hospitals, Thürmann et al.20 observed that the number of drugs included in formularies ranges from some 400 among hospitals with fewer than 500 beds to some 700 among university hospitals, and points to the beta-blocker group and ACE inhibitors as examples of high variability.
These data show that there is considerable variability in the selection and content of drug formularies from different countries and institutions. We also found studies that analyse the causes of variability and factors that influence it, including the weight of efficacy and safety evidence,21–23 economic evaluations,24–28 evaluator perspectives,29,30 availability of institutional guidelines,13,16,27,31 consulted sources of information,32 the promotional role of the pharmaceutical industry,26 etc. Our study was not designed to identify the causes of variability, which are probably similar to those cited in other countries.
Data regarding P&TC activity and the variability of drug-incorporation decisions show that it would be convenient to make use of standard methodology and online cooperation with the SEFH's GENESIS working group. There can be no doubt that the drug assessment, selection and positioning processes would benefit from institutional initiatives, and hospital P&TCs should play an important role in these initiatives due to their presence, structure, procedures and practical experience.
FundingThis project received funding from the Fondo de Investigación Sanitaria del Instituto de Salud Carlos III (health research fund of the Carlos III Health Institute, dependent on the Ministry of Health and Social Policy) in the 2006 call for proposals as Project PI061546.
Conflict of InterestThe authors have no conflicts of interest to declare.
We would like to thank our colleagues at the 175 hospitals that participated in the different phases of the study for the information and completed questionnaires which they provided.
We would also like to thank Teresa Requena Caturla at Hospital Universitario La Paz, Juan Pablo Ordovás Baines at Hospital General Universitario de Alicante, María Ángeles Porta Sánchez at Complejo Universitario Juan Canalejo, José María Recalde Manrique at Centro Andaluz de Documentación e Información de Medicamentos, and Cecilia Calvo Pita from Servicio de Salud de Illes Balears, for their contributions.
Our cordial thanks also go out to the technicians at the research and methodology support unit (UAIM) at Granada's Escuela Andaluza de Salud Pública (EASP, Andalusian school of public health): Elena Corpas Nogales, Leticia García Mochón, and María Moya Garrido.
Please cite this article as: Puigventós Latorre F, et al. Variabilidad en la actividad y los resultados de la evaluación de nuevos medicamentos por las comisiones de farmacia y terapéutica de los hospitales en España. Farm Hosp. 2011;35:305–14.