To analyze the errors in the preparation of parenteral nutrition in a Pharmacy Service, detected through an already consolidated gravimetric and product quality control, and compare them with those detected during the initial years of implementing this quality control.
MethodsAll errors detected through quality control in the compounding of pediatric and adult parenteral nutritions between 2019 and 2021 were prospectively analyzed. This quality control consisted of 3 sequential processes: a visual check, a gravimetric control, and a product control. Errors were classified as gravimetric, when the nutrition had a deviation of more than 5% from the theoretical weight, or as product errors when a qualitative or quantitative error was detected upon reviewing the remainder of the components used. These errors were analyzed in terms of type and the component involved. A comparison was made with the errors detected during the implementation phase of this quality control from 2016 to 2018.
ResultsA total of 41,809 parenteral nutritions were reviewed, and 345 errors were detected (0.83% of the preparations); of these, 59 errors were found in pediatric nutritions (0.68% of them), and 286 in adult nutritions (0.86% of them). Among these errors, 193 were of gravimetric nature, while 152 were detected through product control. The main components involved in product errors were electrolytes, primarily due to the addition of excessive volumes and the use of incorrect components. A significant absolute reduction of 0.71% (P < .05) in the total number of errors was observed when compared to the implementation phase. This reduction was consistent in both gravimetric errors (−0.59%) and product-related errors (−0.12%) (P < .05).
ConclusionsComprehensive quality control of parenteral nutrition preparation is an easily implementable tool that effectively detected and prevented significant errors. Furthermore, its widespread adoption contributed to a reduction in the overall error count.
Analizar los errores de elaboración de las nutriciones parenterales en un Servicio de Farmacia, detectados mediante un control de calidad gravimétrico y de productos ya consolidado, y compararlos con los detectados durante los primeros años de implantación de este control de calidad.
MétodosSe analizaron prospectivamente todos los errores detectados mediante el control de calidad en la elaboración de nutriciones parenterales pediátricas y adultas entre 2019–2021. Este control de calidad comprendió 3 procesos secuenciales: un control visual, uno gravimétrico y uno de productos. Los errores se clasificaron como gravimétricos, cuando la nutrición presentaba una desviación mayor al 5% del peso teórico, o de producto, cuando al revisar el remanente de los componentes utilizados se detectaba algún error cualitativo o cuantitativo. Los últimos se analizaron según el tipo y componente implicado. Se realizó una comparación con los errores detectados durante la fase de implementación de este control de calidad, entre 2016–2018.
ResultadosSe revisaron 41.809 nutriciones parenterales, detectándose 345 errores (0,83% de las nutriciones), 59 en nutriciones pediátricas (0,68% de las mismas) y 286 en nutriciones adultas (0,86% de estas). Un total de 193 errores fueron de tipo gravimétrico, mientras que 152 se detectaron mediante el control de productos. Los electrolitos fueron los principales componentes implicados en los errores de productos, que consistieron mayoritariamente en añadir volúmenes en exceso de los mismos y en el uso de un componente incorrecto. Se encontró una reducción absoluta del 0,71% (p < 0,05) del total de errores en comparación con la fase de implementación. Esta reducción fue consistente, tanto en errores gravimétricos (−0,59%), como en errores de productos (−0,12%) (p < 0,05).
ConclusionesEl control de calidad completo de la elaboración de nutriciones parenterales es una herramienta de fácil implementación que detectó y previno errores relevantes. Además, la consolidación del uso del mismo redujo el número total de errores.
Parenteral nutritions (PNs) are essential in circumstances where enteral feeding is contraindicated, but they are not harmless drugs. The Institute for Safe Medication Practices (ISMP) has categorized them as high-risk medication due to the potential for fatal consequences resulting from incorrect use.1,2
PNs can be found as commercial bags or prepared within Pharmacy Services.3,4 Several stages during their use could be a source of error: prescription, pharmaceutical validation, processing, labelling, and administration.5 It is essential to detect these errors in order to prevent them from reaching the patient, implementing strong quality controls. The compounding process in hospitals is particularly critical, and it should be centralized in the Pharmacy Services and developed by trained personal in specific compounding areas.6 It is essential to ensure the proper selection, volume, and addition order of the components, as well as the absence of microbiological contamination of the preparation. This process is even more crucial preparing infant PNs, and more specifically neonatal PNs, where very small volumes are used and the errors could be really serious.7,8
Several errors have been reported during the compounding process that have had fatal consequences, most of them due to confusion about the products used or the required doses.1 Guenter et al. concluded that errors during the compounding process are one of the main sources of errors in the whole process of using a PN.8 In addition, they indicated that it is 2–3 times more likely to make an error when preparing a PN than another sterile preparation.
In Spain, there is a “Spanish Consensus on the preparation of parenteral nutrient mixtures” which indicates the quality control that should be carried out on PN: visual inspection, gravimetric control, control of the products used, physicochemical analysis of the PN molecules and ions, and microbiological control.4 Other societies, such as the American Society for Parenteral and Enteral Nutrition (ASPEN), prioritize the automation of PN elaboration together with gravimetric controls,2,5 and several authors have recommended the use of refractometry or laboratory techniques (electrophoresis, enzymatic reactions, conductivity, etc.).1,9–12
The Spanish Consensus on the preparation of parenteral nutrient mixtures describes how to perform the gravimetric control process, comparing a theoretical weight calculated using densities of the components and the real weight. However, the maximum limit of this deviation is not consensual: the European Medicines Agency (EMA) sets a maximum deviation of 5%, while the American Pharmacopeia or the Spanish consensus indicates 5% for preparations of more than 100 ml and suggests 3% for those of less than 100 ml.4,13
In 2016, our Pharmacy Service began to implement a complete quality control of compounded PNs, which included visual inspection of the bag, gravimetric control, and control of the products used. Errors detected during the implementation period, between 2016 and 2018, were described and analyzed.14 Since 2019, this quality control was considered consolidated, and it began to be applied to 100% of the compounded PNs. The current study arises to complete that information, with the objective of performing a complete analysis of the errors detected during this consolidation period, and comparing them with those detected during the implementation period.
MethodsA prospective study was carried out to collect all PNs compounding errors detected by quality control from January 01, 2019 to December 31, 2021 in the Pharmacy Department of a tertiary hospital. Pediatric PNs (PPNs), prescribed to patients aged 0–17 years old, and adult PNs (APNs) were analyzed separately.
PNs were prescribed in an individualized manner, based on physician judgment. Subsequently, a pharmacist validated the prescription and generated the preparation sheet, adjusting macronutrient composition when feasible by utilizing complete vials or bags, depending on the product. A pharmacy technician assembled the necessary products on a tray, and a nurse prepared the nutrition. Gravity preparation was employed when using full vials/bags of high-volume products, while a syringe was used for the rest. Quality control was conducted by a different nurse under pharmaceutical supervision, following the procedures outlined in the publication by Melgarejo et al.14 It comprised 3 sequential processes:
- 1.
Visual control of the mixture. The integrity of the bag was checked, assuring the absence of visible particles.
- 2.
Gravimetric control. A maximum variation of 5% was allowed between the real weight and the theoretical calculated weight, based on the densities of the components. Electronic scales with a sensitivity of 0.01 g connected to the MedicalOne Parenteral® software were used for their measurement. A sensitivity analysis was also carried out, using a 3% limit of deviation in PNs of less than 100 ml.
- 3.
Control of the products used. Both the identity and the remaining volume of each used product were visually checked to ensure that they were correct. If very small volumes were used, making visual inspection unfeasible, the remaining quantity was measured using a syringe, with a 5% maximum deviation accepted.
Detection of an error during gravimetric control was considered always as clinically relevant, so the PN was discarded and product control was not performed. On the other hand, if a product error was detected, the pharmacist in charge assessed whether the PN should have been discarded or not.
The analyzed errors were classified as errors detected by product control or by gravimetric control. Errors detected by product control were also classified according to the nature of the product involved: glucose, amino acids, lipids, electrolytes, vitamins, trace elements, water, or drugs. In addition, the type of error was also recorded as: incorrect product, omitted product, over-added product, or under-added product. On the other hand, for the errors detected by gravimetric control, the deviation from theoretical weight of the PN was recorded.
Errors detected were compared with those documented during the implementation and pilot phase of the process (2016–2018), when quality control measures were only applied to 71.46% of the prepared PNs. PNs elaboration and quality control processes were the same as it was mentioned before. Descriptive statistical analysis and comparison of proportions test were performed using STATA version 16. Calculation of confidence intervals was performed using the Wald method.
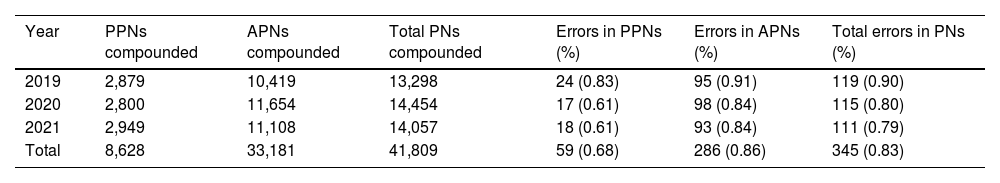
ResultsDuring the 3-year study period, complete quality control was conducted on 41,809 PNs, representing 100% of those compounded. This included 33,181 APNs and 8,628 PPNs. A total of 345 errors were identified (0.83% of total compounded PNs), with 286 occurring in APNs (0.86% of APNs) and 59 in PPNs (0.68% of PPNs) (Table 1).
Distribution of compounded parenteral nutritions and errors detected by quality control, according to year and type of nutrition, adult, or pediatric.
| Year | PPNs compounded | APNs compounded | Total PNs compounded | Errors in PPNs (%) | Errors in APNs (%) | Total errors in PNs (%) |
|---|---|---|---|---|---|---|
| 2019 | 2,879 | 10,419 | 13,298 | 24 (0.83) | 95 (0.91) | 119 (0.90) |
| 2020 | 2,800 | 11,654 | 14,454 | 17 (0.61) | 98 (0.84) | 115 (0.80) |
| 2021 | 2,949 | 11,108 | 14,057 | 18 (0.61) | 93 (0.84) | 111 (0.79) |
| Total | 8,628 | 33,181 | 41,809 | 59 (0.68) | 286 (0.86) | 345 (0.83) |
PPNs: Pediatric Parenteral Nutritions, APNs: Adult Parenteral Nutritions, PNs: Parenteral Nutritions.
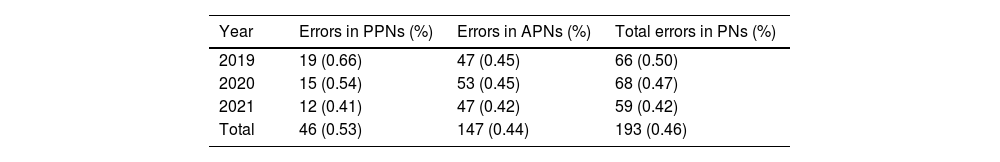
A total of 193 errors were detected through gravimetric control, constituting 0.46% of all PNs, and contributing to 55.94% of the total errors (Table 2). The mean weight deviation from the theoretical weight was 1.79% (95% CI 1.78%–1.81%), while the mean deviation of PNs with gravimetric errors was 9.17% (95% CI 8.09%–10.24%).
Distribution of gravimetric errors according to year and type of nutrition, adult. or pediatric.
| Year | Errors in PPNs (%) | Errors in APNs (%) | Total errors in PNs (%) |
|---|---|---|---|
| 2019 | 19 (0.66) | 47 (0.45) | 66 (0.50) |
| 2020 | 15 (0.54) | 53 (0.45) | 68 (0.47) |
| 2021 | 12 (0.41) | 47 (0.42) | 59 (0.42) |
| Total | 46 (0.53) | 147 (0.44) | 193 (0.46) |
PPNs: Pediatric Parenteral Nutritions, APNs: Adult Parenteral Nutritions, PNs: Parenteral Nutritions.
When analyzing data categorized by the type of nutrition, we identified 147 errors in APNs, representing 0.44% of the compounded APNs. Notably, APNs with errors exhibited an average deviation from their theoretical weight of 9.46% (95% CI 8.39%−10.54%). On the other hand, 46 errors were found in PPNs, constituting 0.53% of the compounded PPNs. Remarkably, PPNs with errors showed an average deviation from their theoretical weight of 8.19% (95% CI 7.11%–9.26%). Average weight for APNs was 1616 g (range: 313–2013 g) and for PPNs was 424 g (range: 34–1330 g).
Of the errors detected, 174 were due to a positive deviation of more than 5% of the real weight compared to the theoretical weight, while 19 errors exhibited a negative deviation of more than 5%.
In PNs of less than 100 ml, taking a 5% deviation limit, 9 PNs (1.14%) were discarded. In a sensitivity analysis adopting a 3% limit, 148 PNs (18.78%) would have been discarded.
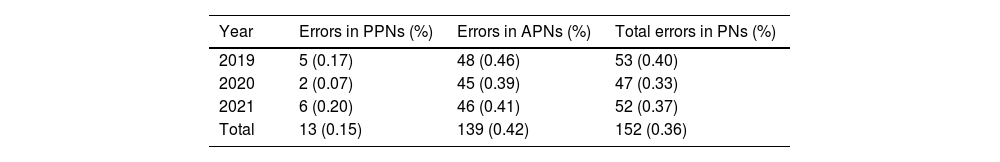
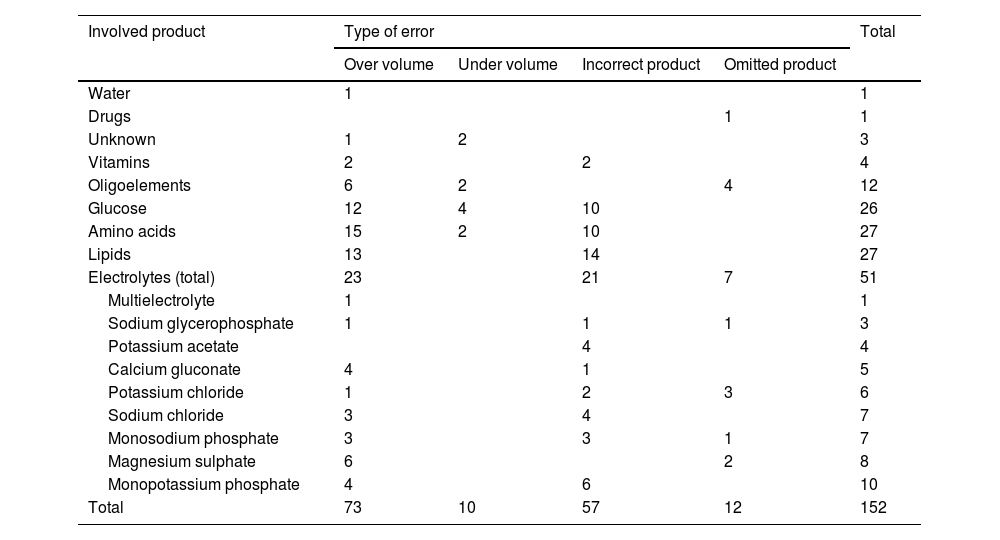
Product controlA total of 152 errors were identified through product control, representing 0.36% of PNs and contributing to 44.06% of the total errors (Table 3). Among these, 139 errors were associated with APNs (0.42% of total APNs), while 13 were related to PPNs (0.15% of total PPNs). Table 4 provides information on the type of error and the associated product. Electrolytes were the most frequent product involved (33.55% of the errors), so this group was detailed. The majority of errors were attributed to an over-added product (48.03%). Insulin was the only medication that was involved in errors.
Distribution of product errors according to year and type of nutrition, adult, or pediatric.
| Year | Errors in PPNs (%) | Errors in APNs (%) | Total errors in PNs (%) |
|---|---|---|---|
| 2019 | 5 (0.17) | 48 (0.46) | 53 (0.40) |
| 2020 | 2 (0.07) | 45 (0.39) | 47 (0.33) |
| 2021 | 6 (0.20) | 46 (0.41) | 52 (0.37) |
| Total | 13 (0.15) | 139 (0.42) | 152 (0.36) |
PPNs: Pediatric Parenteral Nutritions, APNs: Adult Parenteral Nutritions, PNs: Parenteral Nutritions.
Type and amount of product errors, with electrolytes detailed.
| Involved product | Type of error | Total | |||
|---|---|---|---|---|---|
| Over volume | Under volume | Incorrect product | Omitted product | ||
| Water | 1 | 1 | |||
| Drugs | 1 | 1 | |||
| Unknown | 1 | 2 | 3 | ||
| Vitamins | 2 | 2 | 4 | ||
| Oligoelements | 6 | 2 | 4 | 12 | |
| Glucose | 12 | 4 | 10 | 26 | |
| Amino acids | 15 | 2 | 10 | 27 | |
| Lipids | 13 | 14 | 27 | ||
| Electrolytes (total) | 23 | 21 | 7 | 51 | |
| Multielectrolyte | 1 | 1 | |||
| Sodium glycerophosphate | 1 | 1 | 1 | 3 | |
| Potassium acetate | 4 | 4 | |||
| Calcium gluconate | 4 | 1 | 5 | ||
| Potassium chloride | 1 | 2 | 3 | 6 | |
| Sodium chloride | 3 | 4 | 7 | ||
| Monosodium phosphate | 3 | 3 | 1 | 7 | |
| Magnesium sulphate | 6 | 2 | 8 | ||
| Monopotassium phosphate | 4 | 6 | 10 | ||
| Total | 73 | 10 | 57 | 12 | 152 |
132 of the PNs with product errors were discarded (86.84%), while 20 were still used because those errors were considered not clinically relevant.
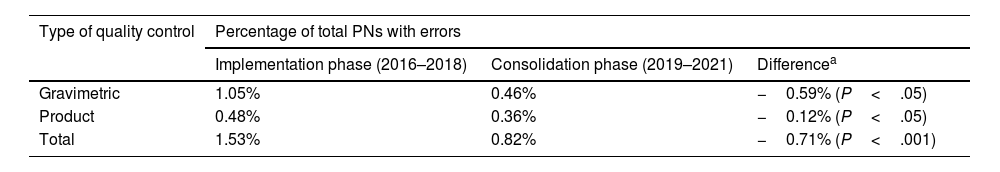
Comparison of errors between implementation and consolidation phasesDuring the implementation phase (2016–2018), quality control was conducted on 28 761 PNs, which accounted for 71.46% of those prepared during this period. A total of 440 errors were detected (1.52%): 1.05% of PNs exhibited gravimetric errors while 0.48% displayed product errors.
When comparing these proportions with the consolidation period, a difference of −0.71% (95% CI −0.56% to −0.85%) was observed in favor of the latter, with an OR of 0.39 (95% CI 0.38–0.40). This difference remained consistent within both subgroups, being −0.59% (95% CI −0.72% to −0.47%) for gravimetric errors and −0.12% (95% CI −0.20% to −0.03%) for product errors. Table 5 shows these differences. Gravimetric errors exhibited reductions in APNs (0.60% vs 0.44% of the total APNs), but especially in PPNs (2.20% vs 0.53% of the total PPNs). Electrolytes remained the primary component involved in product errors.
Comparison of percentage of parenteral nutrition with detected errors, between implementation and consolidation phases.
| Type of quality control | Percentage of total PNs with errors | ||
|---|---|---|---|
| Implementation phase (2016–2018) | Consolidation phase (2019–2021) | Differencea | |
| Gravimetric | 1.05% | 0.46% | −0.59% (P<.05) |
| Product | 0.48% | 0.36% | −0.12% (P<.05) |
| Total | 1.53% | 0.82% | −0.71% (P<.001) |
PNs: Parenteral Nutritions.
This study emerged from the necessity to complete the data collected during the initial years of implementing PN quality control, aligning with the recommendations of the Spanish consensus.4 During the 3 analyzed years, complete quality control was applied to 100% of the personalized PNs prepared, so the process could be considered consolidated.
To the best of our knowledge, we are analyzing one of the largest datasets of compounded PNs. Both the number of PNs performed and the number of errors detected were distributed in a balanced way over the 3 years. It is difficult to find publications to compare these results, since there are very few research that focus exclusively on errors during the compounding process. MacKay et al. analyzed errors at any stage of pediatric parenteral nutrition use (prescription, transcription, preparation, administration)15 and they found errors in 0.27% of them, a percentage much lower than ours (0.68% of the PPNs). Their elaboration process was different: they used automated systems, which could favor the absence of errors, and they used quality controls based on refractometry and laboratory analysis of ions. Another American study found that 0.38%16 of analyzed PNs had errors during preparation, less than half of our findings (0.83%). On the other hand, the ASPEN reported that 37% of the PN manually elaborated had errors, a very high number that could not be comparable to ours because it also included errors in the aseptic technique.8
In our study, errors detected by gravimetric control were 21.24% greater than those detected by product control. This difference can be explained by the fact that product control was not applied to PNs with erroneous weights, which were immediately discarded. Of the PNs, 0.46% exceeded the 5% deviation limit, mostly due to excess weight. These suggested that errors primarily occurred when personnel mistakenly added entire vials/bags of large-volume products when only a fraction was required, such as using a 250 ml vial of 70% glucose to obtain 200 ml of solution. On the other hand, errors in PPNs were more prevalent compared to APNs (0.44% vs 0.53%). This was expected, since PPNs involves smaller volumes, where even a minor measurement error can result in a substantial deviation, remarking how easily this can have repercussions on patients' health.
Very few other researches have focused on the study of gravimetric errors. Serrano et al.17 found that 0.95% of the prepared nutritions have a gravimetric error of more than 5%, which is higher than our findings (0.46%). If we analyze deeply their study, 1.2% of the large-volume nutritions they prepared had an unacceptable gravimetric error, which was 3 times the 0.44% that we obtained in adult PNs. However, they did not report errors in small-volume preparations, although their sample was much smaller than ours (42 weighed PNs), making it difficult to interpret the finding.
It is also noteworthy to analyze low-volume nutritions (<100 ml) sensitivity analysis. The results indicate that if a 3% deviation limit were employed, the percentage of mixtures that would have required disposal would have significantly increased from 1.14% to 18.78%, which is a substantial escalation. However, considering that product control was executed and would have detected any significant errors in the various components of the PN, the 5% limit recommended by the European Medicines Agency4 appears to be appropriate for ensuring safety while optimizing cost-effectiveness. In scenarios where product control is not conducted, implementing a 3% limit could be regarded as a compensatory safety measure.
Product control distinguishes error sources, unlike gravimetric control. In our study, most errors stemmed from over-volume addition and incorrect product use. Over-volume addition often resulted from using fractionated vials/ampoules for low-volume products, inadvertently adding the entire contents. Similar packaging and names for components, coupled with supply shortages, contributed to product selection errors when preparing PNs trays.18 Notably, as it had occurred during the implementation phase, errors related to electrolytes were the most frequently encountered, reflecting all these situations. Electrolyte errors typically involved excess volume due to ampoule fractionation for dosage adjustment. Furthermore, the resemblance of names like monopotassium, monosodium, and dipotassium phosphate, or sodium and potassium chlorides, induced confusion. These formulations were also often affected by supply shortages, leading to substitution with less familiar commercial presentations for the personnel. Unfortunately, we did not find other studies to compare our data.
Another advantage of product control is that it allows rational error evaluation, avoiding unnecessary PN discarding. In our case, 20 erroneous PNs were still used, saving time and resources.
It's worth noting that the percentage of PPNs with product errors is lower than that of APNs (0.15% vs 0.42%), despite PPNs using smaller volumes with a higher potential for errors. This could be explained by the higher number of PPNs that do not pass gravimetric control, resulting in fewer undergoing product control. Additionally, since the implementation of quality control, there has been a strong emphasis on raising awareness of the high risks associated with the preparation of PPNs, which may lead nurses to handle them with greater care than APNs.
Comparing our findings with the quality control implementation phase, we observed a statistically significant 0.71% reduction in error occurrence, with a more pronounced decrease in gravimetric errors (−0.59%) than product errors (−0.12%). This finding showed that the implementation of a quality control system, together with analysis of the errors detected, has resulted in a safer process, probably by raising staff awareness of the importance of compounding NPs and implementing process improvement measures. These results invite other centers to consider implementing a simple and inexpensive quality control system if they do not have one, since it not also prevents errors, but also it seems to reduce them over the time.
Spanish consensus on parenteral nutrition preparation4 also recommends performing physicochemical analyses of PNs: osmolarity, ion content… but these methods are not easy to implement and require specific and expensive equipment. However, simpler methods like capillary electrophoresis for electrolyte detection and enzymatic reactions for glucose detection are fast (less than 10 min) and accurate, and may replace manual controls in the future.11,19–21
Our approach involved manually prepared individualized nutrition, which is vulnerable to human errors at various stages. Automated nutrition preparation is favored in the latest guidelines, as it reduces errors and quality control time by automated detection of nutritional components, although gravimetric control should continue to be performed.4,22 However, implementation costs, barcode reliance, and compatibility with small volumes remain challenges. Finally, other potentials strategies for reducing errors and enhance efficiency involve both the utilization of commercial tricameral bags, into which only trace elements, vitamins, and drugs need to be added, or the standardization of PNs compounding, by exclusively using complete vials/bags, following a dose-binding strategy. Nevertheless, further research is needed to assess non-inferiority in terms of health outcomes compared to completely individualized prescription, as the nutritional intake requirements would be approximate rather than exact.22–25
In conclusion, a complete quality control process for PN elaboration detects potentially serious errors and prevents these from reaching the patient. Both gravimetric and product analyses suggest that the improper fractionation of various dosage forms during the compounding process constitutes a primary error source, often resulting in the addition of the entire volume instead of the intended fraction. Moreover, the implementation of quality control appears to reduce the overall error rate, probably by increasing awareness among the involved personnel and encouraging the implementation of safety strategies.
Ethical considerationsThe study was conducted in accordance with the Helsinki Declarations. Informed consent was not required as no patients were involved.
FundingNo funding.
CRediT authorship contribution statementDaniel Gómez-Costas: Writing – review & editing, Writing – original draft, Software, Project administration, Methodology, Investigation, Formal analysis, Data curation, Conceptualization. Rosa María Romero-Jiménez: Supervision, Methodology, Conceptualization. Maria Elena Lobato-Matilla: Methodology, Conceptualization. Raquel Culebras: Investigation. Judy Alejandra González: Investigation. Sergio Herrero-Bermejo: Visualization. Ana María Herranz-Alonso: Resources, Project administration. María Sanjurjo: Resources, Project administration.
Contribution to the scientific literatureIt elaborates in detail on the errors detected during the production of parenteral nutrition and suggests that quality control not only detects but also reduces errors.
The results encourage the continued use of simple but effective quality control systems in the production of parenteral nutrition.
Conflict of interestNo conflict of interest. The authors have no relevant affiliations or financial involvement with any organisation or entity with a financial interest or financial conflict with the topic or materials discussed in the manuscript. This includes employment, consultancies, honoraria, stock ownership, expert testimony, grants, or patents received or pending.
AcknowledgementsTo the Pharmacy service of the Gregorio Marañón for support in the project.