Improving understanding of actual pulmonary hypertension (PH) treatment adherence patterns is crucial to properly treating these patients. We aimed to primarily assess adherence to treatments used for pulmonary arterial hypertension (PAH) and chronic thromboembolic pulmonary hypertension (CTEPH) specific therapies, identify potential factors related to it and secondly describe its treatment patterns.
MethodsA 6-month observational cross-sectional study in a tertiary care hospital was conducted. Patients with PH-targeted therapy who picked it up in the ambulatory hospital pharmacy and who had been on treatment with the same drug for at least 1 year were included. Adherence was assessed as: 1) Proportion of days covered (PDC); and 2) Simplified Medication Adherence Questionnaire (SMAQ). PDC ≥80% was considered adherent. Statistical analyses were performed to evaluate the study outcomes. Logistic regressions were estimated to identify the association between baseline characteristics and factors associated with adherence. P < 0.05 indicated statistical significance.
ResultsA total of 63 patients with 127 different treatments were included, 71.4% were females with a mean age (SD) of 59 (15) years. PAH was the most common diagnosis (74.6%). Double therapy was used in 39.7% of patients, being the combination of Macitentan + Tadalafil and Ambrisentan + Tadalafil the most prescribed. Endothelin receptor antagonists were the most used treatment (40.2%). Adherence according to PDC was 93.7%, showing no great differences depending on the targeted drug used, and according to SMAQ 61.9%. The agreement degree of both methods was slight (65.1%; Kappa 0.12). Only female sex (OR: 0.23, 95% CI: 0.06–0.90; p = 0.035) was associated with worse adherence in the SMAQ method but not in the PDC. Adverse events were reported by a 55.6% of participants and the perception of effective treatment was high (95.2%).
ConclusionsAdherence to PH therapy differs depending on the assessment method; PDC showed greater adherence rate than SMAQ. According to SMAQ, female sex may have a negative impact on adherence in this cohort, but PDC revealed no factors influencing it. No notable differences in adherence between treatment types were found and generally patients felt the treatments were effective in controlling their disease.
Conocer los patrones de adherencia al tratamiento de la hipertensión pulmonar (HP) es crucial para tratar adecuadamente a estos pacientes. El objetivo principal fue evaluar la adherencia a los tratamientos empleados para la hipertensión arterial pulmonar (HAP) e hipertensión pulmonar tromboembólica crónica (HPTEC), identificar los posibles factores relacionados con la misma y describir los patrones de tratamiento.
MetodologíaEstudio transversal observacional de 6 meses de duración en un hospital terciario. Se incluyeron pacientes que llevaban >1 año con el mismo tratamiento para la HP y lo recogían en farmacia ambulatoria. La adherencia se evaluó mediante: 1) Proporción de días cubiertos (PDC); y 2) Cuestionario simplificado de adherencia a la medicación (SMAQ). Se consideraron adherentes si PDC ≥ 80%. Se emplearon regresiones logísticas para analizar la asociación entre características basales y factores asociados con la adherencia. P < 0,05 se consideró estadísticamente significativo.
ResultadosSe incluyeron 63 pacientes con 127 tratamientos diferentes, el 71,4% eran mujeres con una edad media (DS) de 59 (15) años. La HAP fue el diagnóstico más frecuente (74,6%). La terapia doble se empleó en el 39,7% de pacientes, siendo Macitentan + Tadalafilo y Ambrisentan + Tadalafilo las combinaciones más prescritas. Los antagonistas de los receptores de la endotelina fueron el tratamiento más utilizado (40,2%). La adherencia según PDC fue 93,7%, sin mostrar grandes diferencias entre los fármacos empleados, y según SMAQ del 61,9%. El grado de concordancia de ambos métodos fue bajo (65,1%; Kappa 0,12). Sólo el sexo femenino (OR: 0,23; IC95%: 0,06-0,90; p = 0,035) se asoció a una peor adherencia en el método SMAQ, pero no en PDC. El 55,6% de los participantes experimentaron efectos adversos y la percepción de eficacia del tratamiento fue alta (95,2%).
ConclusionesLa adherencia al tratamiento de la HP difiere según el método empleado. PDC mostró mayor tasa de adherencia mayor que SMAQ. Según SMAQ, sólo el sexo femenino parece tener un impacto negativo en la adherencia, pero la PDC no encontró ningún factor influyente. No hubo grandes diferencias en adherencia entre tipos de tratamientos. En general, los pacientes consideraron que los tratamientos eran eficaces para controlar su enfermedad.
Both pulmonary arterial hypertension (PAH) and chronic thromboembolic pulmonary hypertension (CTEPH) are severe, long-term conditions that call for individualized care. The medical treatment focuses on reducing symptoms, increasing physical work capacity, improving quality of life, and ensuring survival.1 Initially, the pathophysiology of both conditions was poorly understood, and therapeutic advances were slow. However, elucidation of several of the physiological pathways that mediate them has led to the development of a growing repertoire of targeted therapies that have been shown to reduce morbidity and improve functional status in patients with both conditions.2,3
There are several approved medical therapies for PAH and CTEPH: endothelin receptor antagonists, phosphodiesterase type-5 inhibitors, soluble guanylate cyclase stimulators, prostacyclins, and prostanoids.4 These therapies vary in their target pathway, administration route (oral, inhaled, subcutaneous and intravenous drugs) and dosing frequency.5,6
Drug adherence is usually associated with better health outcomes, yet many studies suggest that poor drug adherence is prevalent. According to the World Health Organization (WHO), adherence for individuals with chronic diseases is around 50%.1,5,6 Medication adherence and persistence are critical issues for managing chronic diseases. In patients with chronic medical conditions, several factors may affect medication adherence. These include treatment-related factors (e.g., regimen complexity, toxicity), patient-related factors (e.g., polypharmacy), physician-related factors (e.g., provider-patient communication) and health system-related factors (e.g., financial cost). Factors associated with PAH and CTEPH medication adherence remain controversial.7
To date, there is little understanding of the patterns of adherence to PAH and CTEPH targeted specific therapy.4,7 The factors reported in the studies vary, depending on the method used to assess adherence.1,5,8–10 The integration of a specialty pharmacist as patients care partners has shown to correlate with increased treatment adherence in this population.8 Improving our understanding of real-world adherence patterns of pulmonary hypertension (PH) treatments is essential for providing better care for these patients. The evaluation of factors related to low medication adherence may help guide pharmacists' targeted interventions in this population. Thus, this study aimed to primarily assess adherence to treatments used for PAH and CTEPH specific therapies and identify potential factors related to it and secondly, describe the treatment patterns of both pathologies.
MethodsStudy design and data sourceWe conducted an observational cross-sectional study in a tertiary care hospital from July to December 2023. Data were extracted from the electronic medical records, from the drug dispensing records of the ambulatory pharmacy of the center and from patients' responses to adherence questionnaires. Medical records included demographic data, diagnosis and date of it. Outpatient pharmacy dispensing order included dispensed medications, date of dispensation, quantity dispensed, dose, and number of days' supply. The study complies with the Declaration of Helsinki and was approved by the Ethics Committee of the center (Registration reference HCB/2023/0666). Data from all patients were anonymously collected in an electronic dataset. Written informed consent was provided by all participants.
ParticipantsPatients included in the study were adults aged 18 years or older, with an International Classification of Diseases, 11th Revision Clinical Modification (ICD-11-CM) diagnosis codes for PAH or CTEPH, who picked up targeted medication for it at the outpatient pharmacy of our institution and who had been on treatment with the same drug for at least 1 year or had at least 4 dispensations of the same drug in order to evaluate the adherence. All patients had to be able to write and complete the adherence questionnaire or have a personal caregiver complete it for them. We excluded patients receiving treatment for PAH or CTEPH as off-label or compassionate use.
Medical treatment and analysis of adherenceMedication adherence was measured using two methods:
1) Proportion of days covered (PDC):11 defined as the total days covered by medication fills divided by the total number of days the patient was prescribed the medication during the observation period. To evaluate adherence in the last year, the date of first dispensing was the date of first dispensing one year prior to the date of study's inclusion and the date of last dispensing was taken as the date the patient claimed the drug at the outpatient pharmacy. In patients with more than one treatment, the overall PDC was calculated as the average adherence to all treatments. We used PDC data to predict characteristics associated with adherence and identify patients as adherents based on a PDC threshold greater than or equal to 80%. Given the death of disease-state specific data connecting adherence with clinical outcomes, and its use as a benchmark for adherence in previous trials, this threshold was chosen.8,9
2) SMAQ (Simplified Medication Adherence Questionnaire) questionnaire: It consists of 6 questions that classify patients into non-adherents if they answer yes to the questions “Do you ever forget to take your medication?”, “Do you ever stop taking your medication if you feel unwell?”, “Do you forget to take your medication over the weekend?” Patients are also considered not adherent if they answer no to the question: “Do you always take your medication at the prescribed time?” or if their answer to the question "Since the last visit, on how many days have you not taken the full dose for that day?" is that they missed more than 2 doses in the last week, or that they have not taken all daily doses for more than 2 consecutive days in the last 3 months. It can also be used semi-quantitatively based on the response to “In the last week, how many times did you miss a dose?”. Participants completed the SMAQ questionnaire either during the pharmacy dispensation or at home and then returned it.
As specific targeted medication we included: 1) endothelin receptor antagonists (ERA): ambrisentan, bosentan, macitentan; 2) phosphodiesterase type-5 inhibitors (PDE-5i): tadalafil and sildenafil; 3) soluble guanylate cyclase stimulators (SGCs): riociguat; prostacyclins receptor agonists (PRA): selexipag and 5) prostanoids (PRO): epoprostenol, treprostinil and iloprost.
To describe the characteristics of the study population, we collected the age, gender, main diagnosis, specific targeted medication, its use in monotherapy or combination therapy and the use of concomitant chronic treatments. The following variables were investigated as predisposing factors that could predict adherence: sex, age, cohabitation, level of education, current employment status, time since diagnosis of the disease, number of drugs for other comorbidities, perception of adverse effects and efficacy of the treatment for disease control.
Statistical methods and data managementDescriptive statistics were used to characterize the data. Categorical variables were presented as numbers and percentages, whereas continuous variables were presented as means and standard deviations (SD) or median and interquartile range (IQR). To uncover the predictors of adherence, two different univariate binary logistic regression analyses were conducted (calculating odds ratios with 95% confidence intervals): one for the PDC method and another for the SMAQ questionnaire. The two different binary measures of adherence were used as dependent variables in logistic regressions with demographic and patient-related factors used as predictor variables. All variables collected were introduced into the model as binary independent variables. The statistical criteria for acceptance of variables in the model was statistical significance at p < 0.05. We evaluated the degree of agreement between the two methods according to the kappa index and following the interpretation of Landis & Koch.12 All analyses were performed using Stata®/BE 17.0 software.
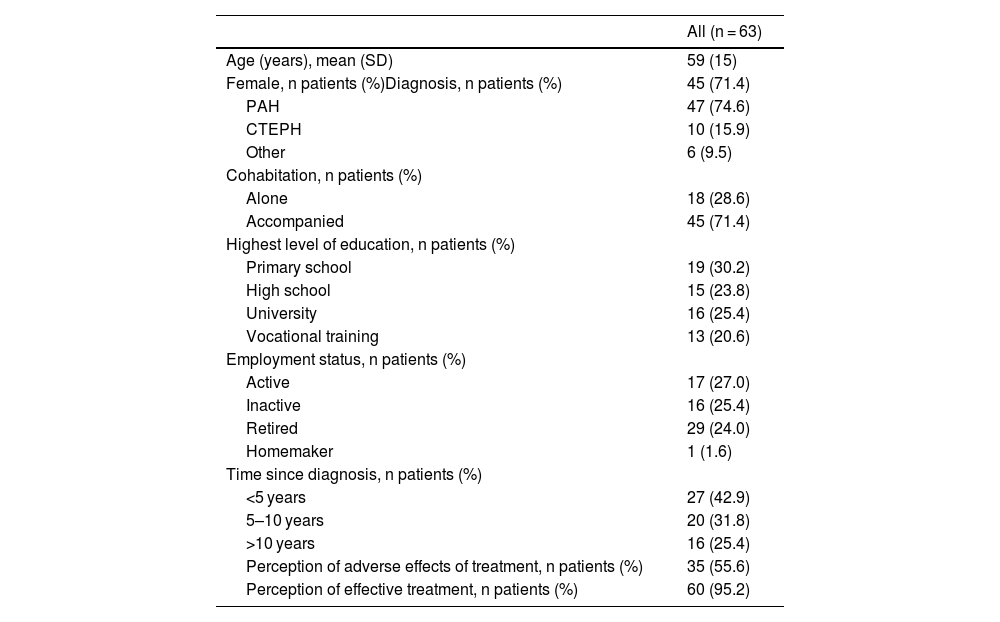
ResultsA total of 63 patients were included in the study. The mean age (SD) was 59 (15) years, 71.4% were females and 74.6% were diagnosed with PAH; 15.9% with CTEPH and 9.5% had other diagnosis. The 55.6% of participants perceived that the treatment caused them adverse effects, with headache and diarrhea being the most reported, and 95.2% of patients felt that the treatment received was effective in controlling their disease. Table 1 summarizes the baseline demographics and the evaluated factor influencing treatment adherence.
Baseline demographics and characteristics of the study population.
| All (n = 63) | |
|---|---|
| Age (years), mean (SD) | 59 (15) |
| Female, n patients (%)Diagnosis, n patients (%) | 45 (71.4) |
| PAH | 47 (74.6) |
| CTEPH | 10 (15.9) |
| Other | 6 (9.5) |
| Cohabitation, n patients (%) | |
| Alone | 18 (28.6) |
| Accompanied | 45 (71.4) |
| Highest level of education, n patients (%) | |
| Primary school | 19 (30.2) |
| High school | 15 (23.8) |
| University | 16 (25.4) |
| Vocational training | 13 (20.6) |
| Employment status, n patients (%) | |
| Active | 17 (27.0) |
| Inactive | 16 (25.4) |
| Retired | 29 (24.0) |
| Homemaker | 1 (1.6) |
| Time since diagnosis, n patients (%) | |
| <5 years | 27 (42.9) |
| 5–10 years | 20 (31.8) |
| >10 years | 16 (25.4) |
| Perception of adverse effects of treatment, n patients (%) | 35 (55.6) |
| Perception of effective treatment, n patients (%) | 60 (95.2) |
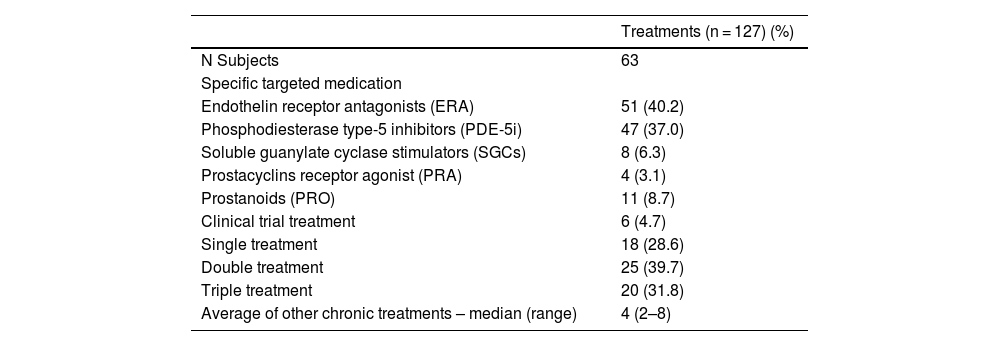
The 63 participating patients had a total of 127 different treatments. The most common type of treatment was the ERA (40.2%), followed by PDE-5i (37.0%). The 39.7% of patients were on double and the 31.8% on triple therapy. In addition, the median number of concomitant treatments for other pathologies was 4 (2–8). Table 2 summarizes the PH-specific treatment patterns.
Pulmonary hypertension-specific treatment patterns.
| Treatments (n = 127) (%) | |
|---|---|
| N Subjects | 63 |
| Specific targeted medication | |
| Endothelin receptor antagonists (ERA) | 51 (40.2) |
| Phosphodiesterase type-5 inhibitors (PDE-5i) | 47 (37.0) |
| Soluble guanylate cyclase stimulators (SGCs) | 8 (6.3) |
| Prostacyclins receptor agonist (PRA) | 4 (3.1) |
| Prostanoids (PRO) | 11 (8.7) |
| Clinical trial treatment | 6 (4.7) |
| Single treatment | 18 (28.6) |
| Double treatment | 25 (39.7) |
| Triple treatment | 20 (31.8) |
| Average of other chronic treatments – median (range) | 4 (2–8) |
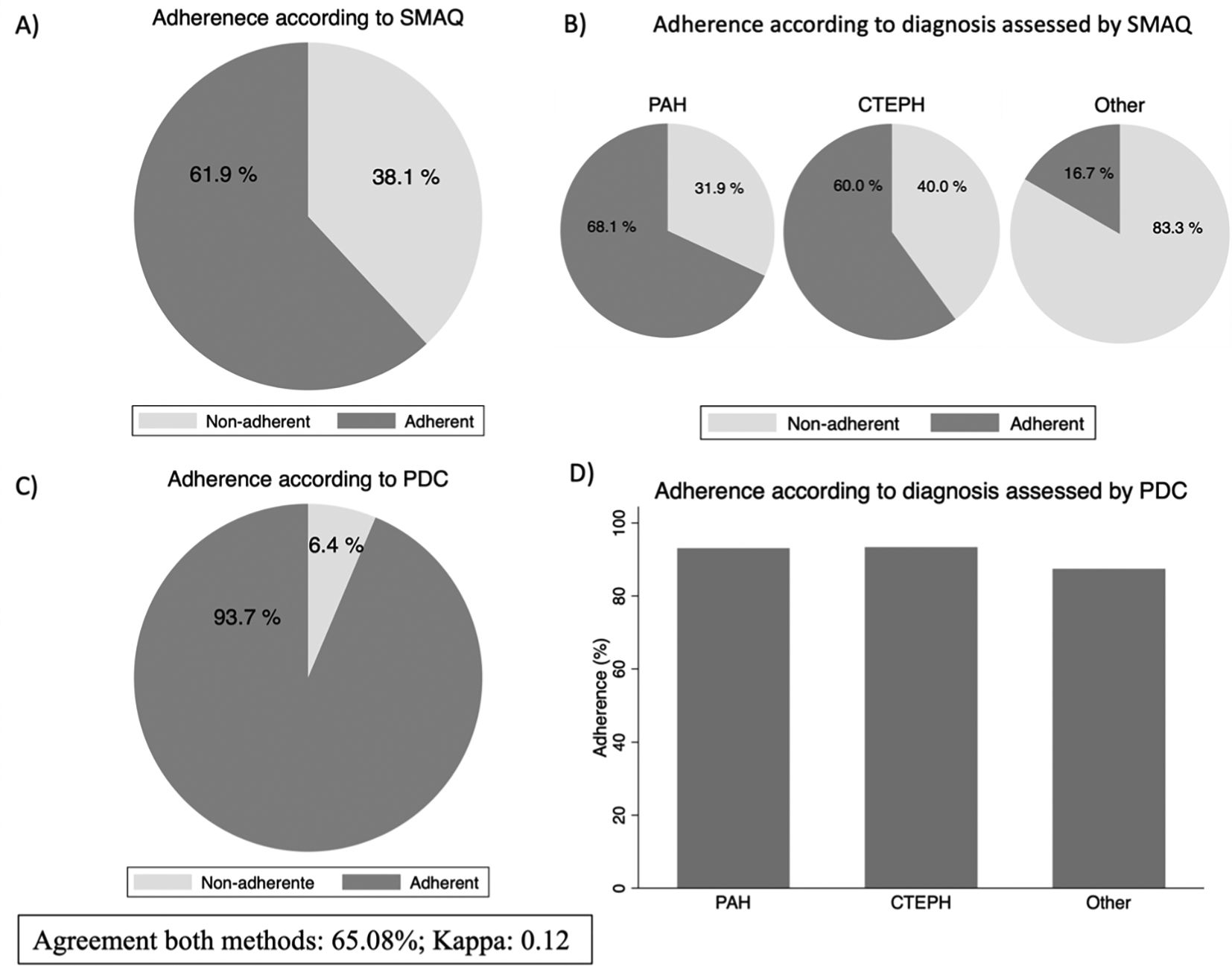
Adherence for the whole population was 61.9% using the SMAQ questionnaire, 68.1% for PAH; 60% for CTEPH and 16.7% for others. The mean doses forgotten in the last week by the non-adherents were 1.75 (2.0) doses. The percentage of adherent patients (≥80%) according to PDC method was 93.7% for all, 93.1% for PAH; 93.4% for CTEPH and 87.4% for others. The percentage of agreement between the two methods was 65.1%; Kappa 0.12, interpretated as slight by Landis & Koch (Fig. 1). The mean adherence (SD) according to PDC for patients with single, double, and triple treatment was 92.8%, (6.9), 93.8% (5.1) and 90.3% (14.5), respectively.
Proportion of adherence assessed by SMAQ questionnaire (A); adherence by diagnosis according to SMAQ questionnaire (B); adherence assessed by PDC (C); Adherence by diagnosis according to PDC (D).
Abbreviations: CTEPH: Chronic thromboembolic pulmonary hypertension; PAH: Pulmonary arterial hypertension; PDC: Proportion of days covered.
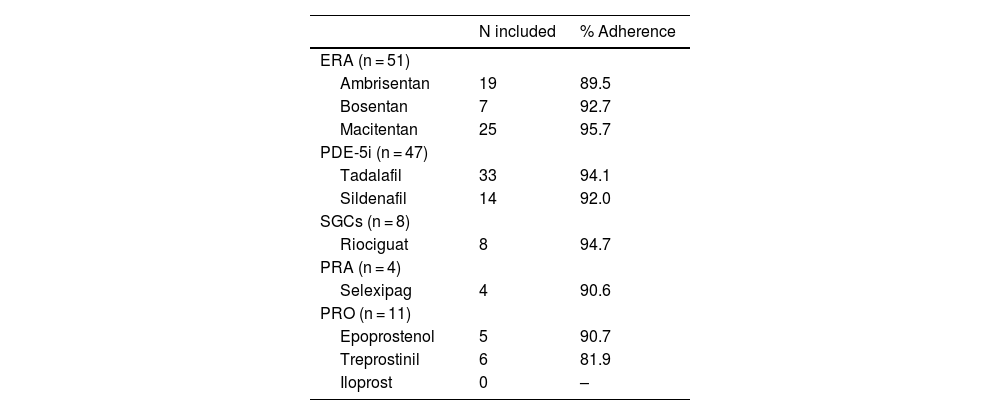
The evaluation of specific adherence to each type of treatment revealed that ERA were the most prescribed treatment, with macitentan (n = 25) being the most used, with a mean adherence of 95.7%. In PDE-5i, tadalafil (n = 33) was the most prescribed, with a mean adherence of 94.1%. The other three pharmacological groups were less employed. The medication groups used by participants and their adherence are shown in Table 3. The most used treatment combination was tadalafil + macitentan in 14 patients, followed by tadalafil + ambrisentan in 13 patients.
Adherence to pulmonary hypertension-specific treatment assessed by PDC.
| N included | % Adherence | |
|---|---|---|
| ERA (n = 51) | ||
| Ambrisentan | 19 | 89.5 |
| Bosentan | 7 | 92.7 |
| Macitentan | 25 | 95.7 |
| PDE-5i (n = 47) | ||
| Tadalafil | 33 | 94.1 |
| Sildenafil | 14 | 92.0 |
| SGCs (n = 8) | ||
| Riociguat | 8 | 94.7 |
| PRA (n = 4) | ||
| Selexipag | 4 | 90.6 |
| PRO (n = 11) | ||
| Epoprostenol | 5 | 90.7 |
| Treprostinil | 6 | 81.9 |
| Iloprost | 0 | – |
Abbreviations: ERA: Endothelin receptor antagonists; PDE-5i: Phosphodiesterase type 5 inhibitors; SGCs: Soluble guanylate cyclase stimulator; PRA: Prostacyclins receptor agonist; PRO: prostacyclin therapy.
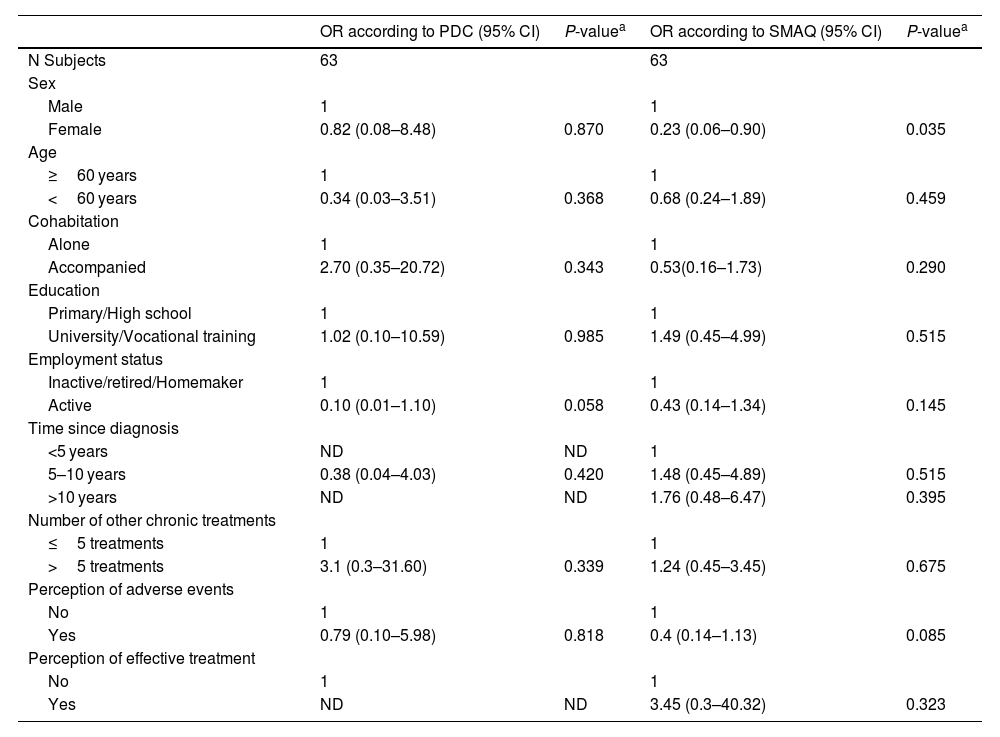
The evaluation of the factors that could influence adherence to PH treatment according to the PDC method, showed that none of them were influent (p > 0.05). In the SMAQ method, only female sex (OR: 0.23, 95% CI: 0.06–0.90; p = 0.035) had a negative impact on adherence. The rest of factors had no influence. The data is summarized in Table 4.
Logistic regression analysis to assess the relationship between adherence and associated factors.
| OR according to PDC (95% CI) | P-valuea | OR according to SMAQ (95% CI) | P-valuea | |
|---|---|---|---|---|
| N Subjects | 63 | 63 | ||
| Sex | ||||
| Male | 1 | 1 | ||
| Female | 0.82 (0.08–8.48) | 0.870 | 0.23 (0.06–0.90) | 0.035 |
| Age | ||||
| ≥60 years | 1 | 1 | ||
| <60 years | 0.34 (0.03–3.51) | 0.368 | 0.68 (0.24–1.89) | 0.459 |
| Cohabitation | ||||
| Alone | 1 | 1 | ||
| Accompanied | 2.70 (0.35–20.72) | 0.343 | 0.53(0.16–1.73) | 0.290 |
| Education | ||||
| Primary/High school | 1 | 1 | ||
| University/Vocational training | 1.02 (0.10–10.59) | 0.985 | 1.49 (0.45–4.99) | 0.515 |
| Employment status | ||||
| Inactive/retired/Homemaker | 1 | 1 | ||
| Active | 0.10 (0.01–1.10) | 0.058 | 0.43 (0.14–1.34) | 0.145 |
| Time since diagnosis | ||||
| <5 years | ND | ND | 1 | |
| 5–10 years | 0.38 (0.04–4.03) | 0.420 | 1.48 (0.45–4.89) | 0.515 |
| >10 years | ND | ND | 1.76 (0.48–6.47) | 0.395 |
| Number of other chronic treatments | ||||
| ≤5 treatments | 1 | 1 | ||
| >5 treatments | 3.1 (0.3–31.60) | 0.339 | 1.24 (0.45–3.45) | 0.675 |
| Perception of adverse events | ||||
| No | 1 | 1 | ||
| Yes | 0.79 (0.10–5.98) | 0.818 | 0.4 (0.14–1.13) | 0.085 |
| Perception of effective treatment | ||||
| No | 1 | 1 | ||
| Yes | ND | ND | 3.45 (0.3–40.32) | 0.323 |
Abbreviations: ND: No data, not possible to calculate the value; PDC: Proportion of days covered; OR: Odds ratio.
This study set out to investigate the treatment patterns and adherence of patients to PH-targeted therapies and explore the medication and patient-related factors that might influence it. The primary findings were that most of the patients were on a double (39.7%) or triple (31.8%) targeted therapy to control its disease and many had other comorbidities that required other chronic treatments (median: 4; IQR: 2–8). Patient's perception of PH-treatments was positive, 95.2% felt they were effective in control the disease although 55.6% reported adverse events. Nevertheless, adherence to the treatments was 93.7% assessed by PDC and 61.9% assessed by SMAQ, which agrees with previous studies.1,5–9,13,14
The most prescribed combination therapy was tadalafil + macitentan and tadalafil + ambrisentan. Our internal protocol for PH treatment recommends the use of combination therapy (ARE + PDE-5i) in intermediate risk patients and low-risk patients without criteria for monotherapy. If the risk is high, a parenteral PRO is added. The upfront combination therapy was recommended based on the results from the AMBITION trial, in which the combination of ambrisentan and tadalafil significantly reduced the time to composite clinical failure compared with monotherapy in naïve patients (hazard ratio (HR), 0.50; 95% CI, 0.35–0.72; p < 0.001).7,15 Current treatment guidelines recommend a combined drug therapy that targets more than 1 biological pathway, (eg, ambrisentan and tadalafil).16 A meta-analysis showed that combined therapy was associated with a significant reduction in clinical worsening compared with monotherapy and highlighted the need to identify novel therapeutic targets for PAH, as many patients still had clinical worsening with combination therapy.17 The therapeutic arsenal for this pathology is expected to increase to improve patients' diagnosis and prognosis.18
Reported adherence of PDE-5i drugs in the treatment of PH, as assessed by PDC, is highly variable, ranging from 94% in the study conducted by Shah NB et al.8 to 47% in the studies conducted by Ruiz et al.13 and Waxman et al.9. In our study, adherence measured by SMAQ was lower than that reported by PDC. In our study, adherence measured by SMAQ was lower than PDC. This method may be stricter in classifying patients as non-adherent, as a single unfavorable response out of the six questions is enough to categorize a patient as non-adherent. However, the PDC method might overestimates adherence since it reflects dispensing but not real consumption. The results of the SMAQ in the group of “other” diagnosis, which includes Raynaud's disease and other forms of PH that require specific targeted therapies, showed a much lower adherence than the rest. Only 1 of the 6 patients (16.7%) included was considered adherent.
In general, adherence measured by PDC was high (>90%). The highest adherence was found for macitentan (95.7%) and the lowest for treprostinil (81.9%). Certain PAH-targeted medications such as sildenafil, riociguat, and inhaled treprostinil are administered three or more times daily, and this can be challenging for patients who may forget to take all doses within the day.5,10 According to the data from this study, adherence was not negatively impacted by higher dose frequency, as riociguat had the second-highest adherence: 94.7% and sildenafil 92.0%. The lower adherence to prostanoids can be explained by the burden of administration and side effects of parenteral and inhaled prostanoids.3
There is no universal method to assess adherence to PH-targeted therapies; PDC has been the most widely employed method,1,3,8,9,13,14,19,20 but other methods like the Morisky Medication Adherence Scale (MMAS-8) have been used.5,6 PDC is an objective measure that is easy to calculate from pharmacy dispensations but reflects prescription refills rather than actual medication consumption.8 Questionnaires as MMAS-8 have the main limitation that adherence is self-reported, and individuals might report higher adherence in those situations.6 In our study we used two methods to evaluate the agreement between both, and we found there is slight agreement between them (65.08%; Kappa 0.12). This could explain why the adherence results between studies are so inconsistent.
Our study found no significant association between adherence and baseline, or clinical characteristics in the logistic regression analysis, except for the female sex (OR: 0.23; 95% CI: 0.06–0.90) in the SMAQ data only, which could be a confounding factor due to the low number of patients in this cohort. Our findings align with several studies reporting age, gender, and socioeconomic status unrelated to medication adherence.1,7–9 Although, according to our results, gender remains controversial. Grady D et al., associated adherence with older age, monotherapy treatment and having a higher number of comorbidities or concurrent medicines.5 While Kjellström B et al., linked good adherence to shorter time since diagnosis in PAH and fewer concomitant other chronic treatments in CTEPH.1 In our study, though nonsignificant, adherence favored older patients, those taking >5 concomitant treatments, longer time since diagnosis and perceiving effective treatment. The positive impact of concomitant medication intake on adherence implies that taking many medications is advantageous. It is possible that if a patient has a medication-taking routine with other medicines, then taking PH-treatments is little extra burden.5 High costs or co-payments for treatment and healthcare contribute to poor medication adherence.1,6,14 However, in this study, those factors were not evaluated as the Spanish healthcare system covers most prescription costs.
It is noteworthy that more than half of the patients (55.6%) perceived adverse events and, despite this, 95.2% reported that it was effective. Headache and diarrhea were the most common, but also myalgia and spider veins in the legs were also reported. Those results agree with previous studies.8 Shah N et al., showed that patients reporting adverse events were more likely to be non-adherent to PDE-5i compared to patients who did not experience it. Patient's perception is an influent factor in adherence and is underreported in HP studies1 Ivarsson B et al, by using the BMQ-S questionnaire, showed that most patients understood the necessity of their medication to maintain or improve their health but almost half of them had concerns about potential adverse consequences of drug intake.6 There are different patient-reported outcome measures (PROMs) that assess PH-specific health-related quality of life, among which the following stand out: Cambridge Pulmonary Hypertension Outcome Review, emPHasis-10, Living with Pulmonary Hypertension Questionnaire, and Pulmonary Arterial Hypertension-Symptoms and Impact.21 The use of PROMs should be implemented more in clinical practice.
It has been demonstrated that involving a clinical pharmacist in the process of care of PAH and CTEPH patients' improves drug access, adherence rates and the coordination of the whole clinical team.8 The clinical pharmacist provides patient education during the dispensations in the ambulatory pharmacy and collaborates with physicians to determine cost-effective and optimal treatment regimens. It is possible that the high adherence rates found in our study are favored by the ongoing care from the healthcare providers and frequent visits to the pharmacy. These patients request their medication every 2–3 months and have a therapeutic follow-up.
To the best of our knowledge, this research is the first study investigating PH-specific treatments patterns and assessing medication adherence among patients in Spain using different methods. Most studies have been conducted in the US3,8,9,13,14,19,20 and Sweden,1,6 which have different healthcare system. Nevertheless, several limitations must be acknowledged when interpreting these results. First, the number of patients was limited, and data came from a single center. It is possible that our findings may not be generalizable to all patients with PH treatments. Second, the proportion of patients with CTEPH was low. Third, numerous potential adherence factors have been identified in the literature, many of which have been difficult to quantify for this study. It's possible that some of these variables may play a role in the observed outcomes. Fourth, in the PDC method we established 80% as the cut-off point, according to what is reported in the literature, but there is no fixed value established. Future studies assessing medication adherence in larger populations would provide meaningful information to confirm our results and determine other possible factors.
In conclusion, this research indicates that adherence to PH-treatment varies according to the method used to assess it 93.7% with PDC and 61.9% with SMAQ, with a light degree of agreement between the two. There are no major differences in adherence between the types of treatment and most patients feel that the treatments are effective in controlling their disease. No clear predictors of poor adherence were found, except for female sex, which had a negative impact but only in the SMAQ method.
Contribution to the scientific literatureAdherence to PH-treatments is generally high, although it differs depending on the assessment method, which may explain the different results obtained in clinical practice. Patients with pulmonary hypertension have a positive view of the treatment, considering it effective in controlling their disease despite the high frequency of adverse effects. Knowing the factors that can influence adherence allows a more patient-centered approach and may help guide pharmacists' targeted interventions in this population. No conditioning factors for poor adherence were found in this cohort. The continuous care offered by healthcare providers may contribute to the high adherence rates obtained in our study.
This study provides valuable information on the characteristics of PH patients from a perspective of great interest to hospital Pharmacists.
Statement of authorshipMarta Albanell Fernández contributed to the design, data collection, interpretation, statistical analysis and writing of the article. Margalida Mestre, Belen López, Rubén Gonzalez, Jaume Planas, Marta Serrano, Emma Wilhelmi, Thais Lizondo contributed to data collection. Dolors Soy contributed to the critical review of the article. Maite Martin contributed to the design, conception, data collection, supervision, and critical review of the article. All authors have approved the final version for publication.
FundingNone declared.
CRediT authorship contribution statementMarta Albanell-Fernández: Writing – review & editing, Writing – original draft, Methodology, Investigation, Formal analysis, Data curation, Conceptualization. María Margalida Mestre: Writing – review & editing, Data curation. Belén López: Writing – review & editing, Data curation, Conceptualization. Rubén González-García: Writing – review & editing, Data curation, Conceptualization. Jaume Planas: Writing – review & editing, Data curation. Marta Serrano: Writing – review & editing, Data curation. Emma Wilhelmi: Writing – review & editing, Data curation. Thais Lizondo: Writing – review & editing, Data curation. Dolors Soy: Writing – review & editing, Validation, Supervision. Maite Martín-Conde: Writing – review & editing, Writing – original draft, Supervision, Project administration, Methodology, Data curation, Conceptualization.