This scoping review aimed to map the evidence of pharmacist-led medication reconciliation in hospital emergency services in Brazil.
MethodWe performed a scoping review by searching electronic databases LILACS, Pubmed, Embase, CINAHL, Scopus, Web of Science, Clinical trials, REBEC e Cochrane and conducting a manual search to identify studies published up to 20 October 2021. Studies that addressed pharmacist-led medication reconciliation in hospital emergency services in Brazil, regardless of clinical conditions, and outcomes evaluated, were included.
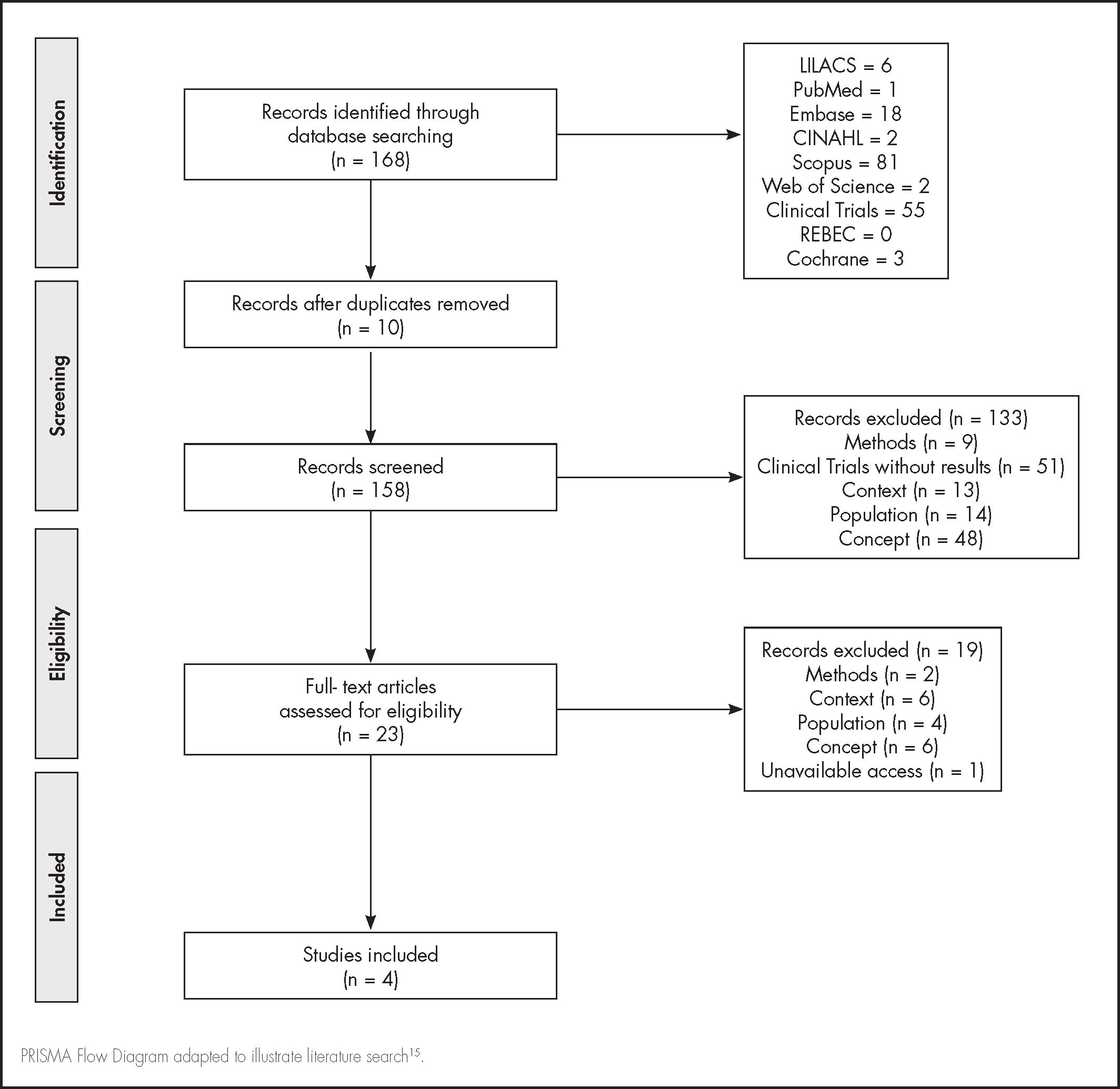
ResultsA total of 168 studies were retrieved, with three matching the inclusion criteria. Most studies performed pharmacist-led medication reconciliation at emergency department admissions, but it was not the primary pharmaceutical attribution in this setting. Medication errors were identified during the medication reconciliation process, being drug omission the most reported. Studies did not describe the concerns in collecting the best medication history from patients and the humanistic, economic, and clinical outcomes of pharmacist-led medication reconciliation.
ConclusionsThis scoping review revealed the lack of evidence about the pharmacist-led medication reconciliation process in the emergency setting in Brazil. The findings suggest the need for future studies in this context.
Documentar la evidencia de la conciliación de medicamentos dirigida por farmacéuticos en los servicios de emergencia hospitalarios en Brasil.
MétodoSe realizó una revisión sistemática exploratoria de bases de datos electrónicas LILACS, Pubmed, Embase, CINAHL, Scopus, Web of Science, Clinical Trials, REBEC y Cochrane para identificar estudios publicados hasta el 20 de octubre de 2021. Los estudios incluidos abordaban la conciliación de medicamentos dirigida por farmacéuticos en los servicios de emergencia hospitalarios en Brasil, independientemente de las condiciones clínicas y los resultados evaluados.
ResultadosSe recuperaron un total de 168 estudios, tres de los cuales cumplieron los criterios de inclusión. La mayoría de los estudios realizaban la conciliación de la medicación dirigida por el farmacéutico en las admisiones al servicio de urgencias, pero ésta no era la principal atribución farmacéutica en ese contexto. Los errores de medicación fueron identificados durante el proceso de conciliación de medicamentos, siendo la omisión de medicamentos el error más reportado. Los estudios no hacían referencia a la importancia de recabar un historial farmacológico lo más completo posible ni a los resultados humanísticos, económicos y clínicos de la conciliación de medicamentos dirigida por farmacéuticos.
ConclusionesEsta revisión sistemática exploratoria reveló la falta de evidencia sobre el proceso de conciliación de medicamentos dirigido por farmacéuticos en los servicios de urgencias de Brasil. Los hallazgos sugieren la necesidad de seguir investigando sobre este asunto.
Medication reconciliation (MR) is encouraged in the third World Health Organization (WHO) Global Patient Safety Challenge: “Medication Without Harm” as a strategy capable of mitigating and preventing drug-related morbidity and mortality by obtaining an accurate and detailed history of pharmacotherapy1. Adverse drug events are the first cause of harm and death related to health care systems, and poor communication between health professionals and between health professionals and patients and carers is a significant factor in their occurrence2.
MR is “the formal process in which healthcare professionals partner with patients to ensure accurate and complete medication information transfer at interfaces of care” and consists of four steps: 1) obtain the best possible medication history (BPMH) by interviewing patients and carers, checking referral letters and other information sources; 2) verify the accuracy of the history with one or more sources; 3) reconcile the BPMH with prescribed medicines and resolve discrepancies with prescriber and document changes; 4) guide patients and carers about the modifications, include a list of current medicines when care is transferred2. This process is recommended to be carried out within 24 hours or sooner if clinically necessary, in acute setting after admission or/ and when the patient moves from one care setting to another, and it is essential to note that the term medicine also refers to over-the-counter medicines and those related to medicine3. MR should investigate these products’ use3.
Patient care starts at the first contact with the health team, which is a critical moment to collect the patients’ medication history to identify medicines used before admission, collaborate with the diagnosis, and optimize treatment safety1. Emergency department (ED) admission medication history obtained by pharmacists are more accurate than other health care team members, and studies suggest that when this professional is involved in gathering and validating the patient's BPMH, there is an impact on preventing medication discrepancies and medications errors (ME) with the potential to cause harm2,4–6. Pharmacists’ involvement in MR is related to a more efficient process, making this an attractive economic strategy to promote patient safety1.
ME and discrepancies are more frequent in ED admissions when comparing MR performed by usual care with pharmacist-led MR, and drug omissions are usually the leading cause of discrepancies4–7. Moreover, pharmacist-led MR in care transitions is associated with a lower rate of ED readmissions7. The global costs associated with ME are estimated at $42 billion per year1. In this context, measures to prevent ME are essential for promoting patient safety, and MR in care transitions can be considered an essential step for ensuring medication safety1,8.
Most evidence quantifies ME, discrepancies, and the potential for patient harm identified with pharmacist-led MR process are from studies in developed countries4–7,9. WHO suggests that these events occur with the same frequency in developing countries1. Such information makes the need for studies about the MR process scenario in these countries, enabling strategies to promote patient safety in care transitions.
Given the importance of the pharmacist's presence in emergency hospital services and their involvement in the MR process, such as the impact on patients’ health and safety, this review sought to identify the evidence related to pharmacist-led MR in emergency hospital services in Brazil.
MethodsStudy designWe conducted a systematic scoping review following the recommendations by the Joanna Briggs Institute (JBI) and the PRISMA-ScR (Preferred Reporting Item for Systematic Reviews and Meta-analyzes for scoping review) statement10,11.
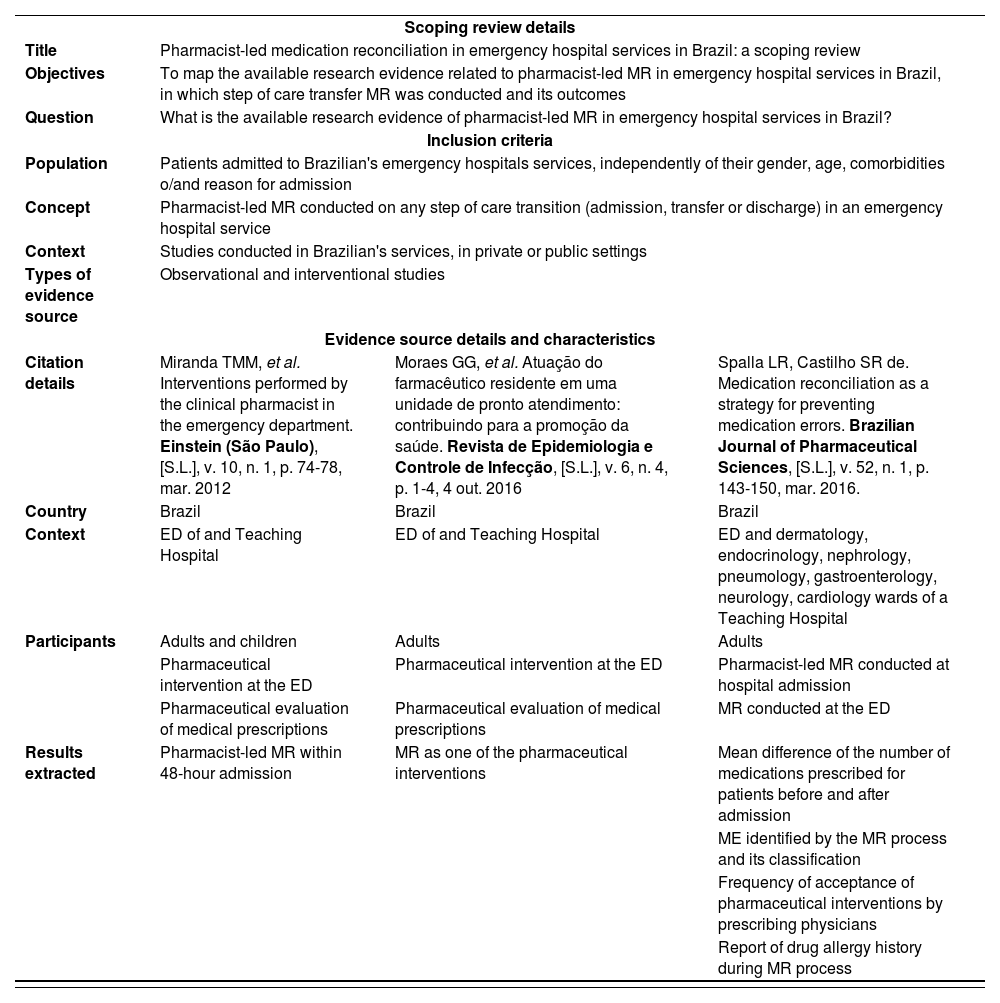
This scoping review's objectives, inclusion criteria, and methods have been specified previously and documented in a protocol available upon request to the corresponding author.
Search strategy and inclusion criteriaWe included Portuguese, English, and Spanish studies published up to October 20, 2021. A preliminary systematic search was performed on the PubMed database on August 22, 2020. The words contained in the titles and abstracts of relevant articles and their respective terms were used to develop the search strategy and adapted, subsequently, for each database used. The last search was on Cochrane, LILACS, PubMed, Embase, CINAHL (Cumulative Index to Nursing and Allied Health Literature), Scopus, Web of Science, Clinical Trials and REBEC (Registro brasileiro de ensaios clínicos) databases on October 20, 2021 (Appendix 1). We explored the reference lists of the included articles to identify relevant articles not retrieved in the electronic searches.
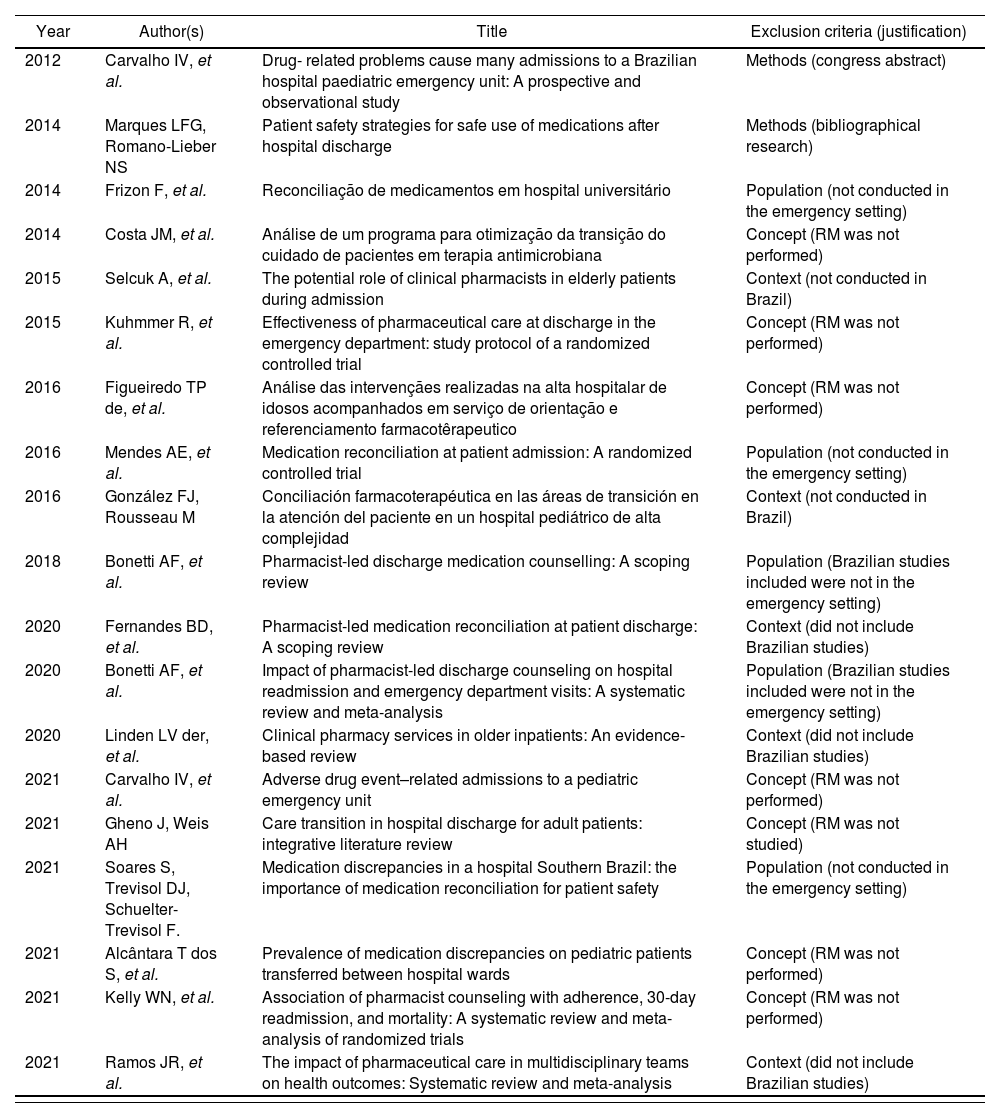
This review included systematic reviews, observational, interventional studies, thesis, and dissertations, with no limit of the publication date, related to patients admitted to emergency hospital services in Brazil submitted to MR by a pharmacist at any stage of care transition, from public or private hospitals, regardless of the outcome assessed. The exclusion criteria were: 1. studies unrelated to Brazilian hospital emergency services; 2. MR performed by other health professionals (not a pharmacist) or by a multidisciplinary team; 3. conferences abstracts, case reports, book chapters and letters to the editor; 4. studies without full-text access.
Eligibility, extraction and presentation of dataWe used the online tool for systematic reviews to select studies, Rayyan QCRI12. We first removed duplicates, and then two independent reviewers examined titles and abstracts to identify studies that met the study's inclusion criteria. After that, both reviewers read the full text of the eligible studies for the final selection. The two reviewers resolved discrepancies by consensus.
The quantitative data from the studies included were extracted and entered into a Microsoft Excel spreadsheet to be analyzed descriptively. The results were presented in a narrative form, with figures and tables. The variables studied were related to the MR process, as well as the characteristics of the included studies, which are: year, location, number of patients and interventions, the study design, objective, the pharmaceutical services described, the time of MR, limitations, conclusions, and findings related to this review.
Systematic scoping reviews aim to map all published evidence of a given subject without excluding studies by analyzing methodological quality10. Thus, following the JBI recommendations, this review did not seek to analyze the methodological quality of the articles included.
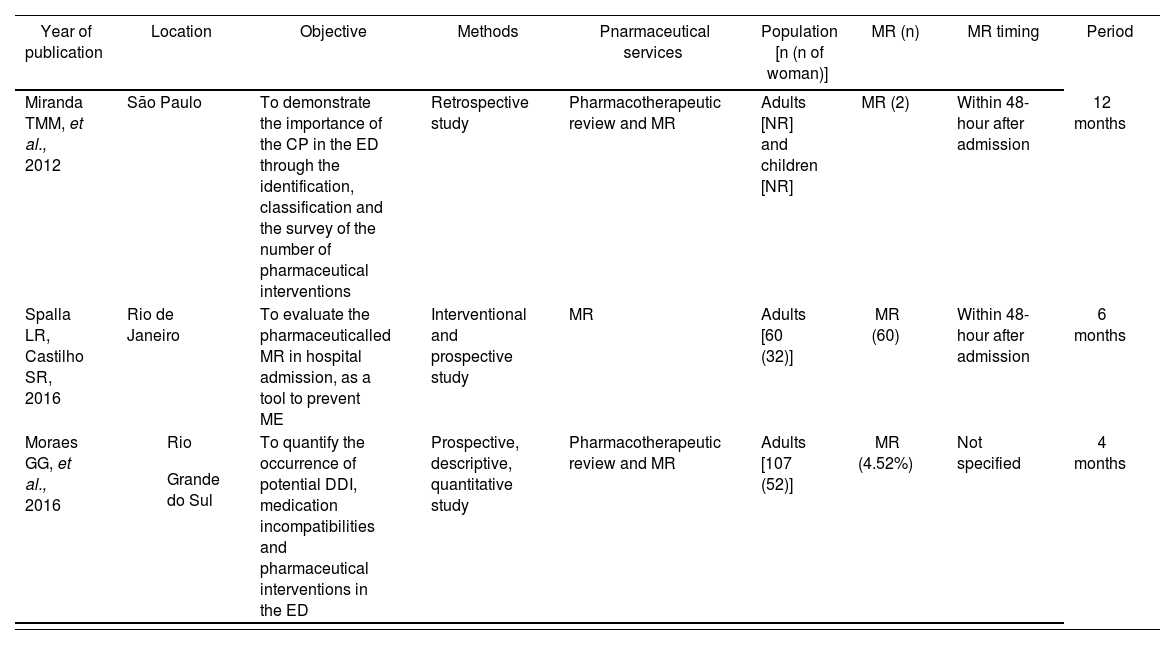
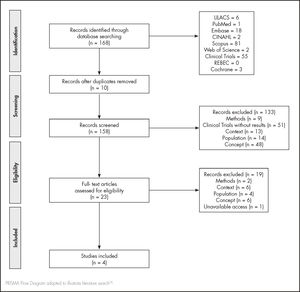
ResultsThe search strategy retrieved 168 articles. After the screening and full-text reading, three articles were eligible for data extraction (Figure 1). The excluded and included studies’ references are available in Appendix 2 and 3, respectively. Table 1 summarizes the main characteristics and results of the included studies.
Main characteristics of included studies.
| Year of publication | Location | Objective | Methods | Pnarmaceutical services | Population [n (n of woman)] | MR (n) | MR timing | Period |
|---|---|---|---|---|---|---|---|---|
| Miranda TMM, et al., 2012 | São Paulo | To demonstrate the importance of the CP in the ED through the identification, classification and the survey of the number of pharmaceutical interventions | Retrospective study | Pharmacotherapeutic review and MR | Adults [NR] and children [NR] | MR (2) | Within 48-hour after admission | 12 months |
| Spalla LR, Castilho SR, 2016 | Rio de Janeiro | To evaluate the pharmaceuticalled MR in hospital admission, as a tool to prevent ME | Interventional and prospective study | MR | Adults [60 (32)] | MR (60) | Within 48-hour after admission | 6 months |
| Moraes GG, et al., 2016 |
| To quantify the occurrence of potential DDI, medication incompatibilities and pharmaceutical interventions in the ED | Prospective, descriptive, quantitative study | Pharmacotherapeutic review and MR | Adults [107 (52)] | MR (4.52%) | Not specified | 4 months |
CP: clinical pharmacist; DDI: drug to drug interactions; ED: emergency department; ME: medication errors; MR: medication reconciliation; NR: not reported. Source: The authors.
The included articles were published between 2012 and 2016, performed in Brazil by pharmaceutical services from university hospitals that assist patients from the Unified Health System (SUS)13,14 and from a private hospital15. The majority of studies were observational13,15, and one study was interventional14, with patients admitted to the ED. One study also included patients admitted for elective procedures but did not describe the admissions rate into the different wards14.
The Spalla and Castilho (2016) study conducted MR for all included patients (n = 60). Moraes et al. (2016) included 107 patients, and of all drug prescriptions analyzed (n = 265), MR was performed in only 4.5% (n = 12) of them. Miranda et al. (2012) did not report the number of patients included but the number of prescriptions analyzed (n = 3,542), leading to 1,238 pharmaceutical interventions, of which 0.16% (n = 2) were MR. Factors such as the lack of information on patients’ drug history, medical records14 and the pharmacist in emergency units for less than 24 hours were limitations for MR process and the pharmacists’ performance in the ED13–15.
In one study, the MR (n = 60) led to the detection of ME (n = 26) related to drug omission (61.50%), followed by dose errors (11.54%) and prescription of the wrong drug (3.85%)14. The other two studies included did not describe results from the pharmacist-led MR13,15.
Most studies reported that the MR process was conducted at hospital admission14,15. For Miranda et al. (2012), MR was the least reported pharmaceutical intervention. The authors justified that the hospital's policy determines that MR may be performed within 48 hours after hospital admission, making this process not a priority in the ED. Other pharmaceutical interventions described as more frequently in the studies were drug-to-drug interactions13 and dose adjustment15 but none was described as related to the MR process.
One study described the MR process through an interview to compare the patients’ previous medication history list with the hospital prescriptions after admission14. None of the studies described the concern with obtaining the BPMH for the MR process, nor its clinical or humanistic outcomes.
DiscussionAccording to our knowledge, this is the first scoping review that maps studies related to pharmacist-led MR in the emergency setting in Brazil. Only three studies met the inclusion criteria, suggesting a scarcity of evidence about this topic. These results reinforce the data presented by the WHO on the absence of evidence on drug safety in care transitions in middle-income countries1. Pharmaceutical care history in Brazil is relatively recent compared to high-income countries16. Publications about the clinical pharmacist activities in Brazil are dated from 200417, and their attributions were first regulated in 201318. Therefore, clinical pharmacy is under development in Brazil, and their activities in the emergency setting are not fully established, which may justify the period of studies publications. Moreover, some barriers described to pharmacist-led MR in the ER are the substantial number of assignments designated to this professional in this setting and the lack of human resources14,15,19.
The studies included in this review pointed to the short period of hospitalization and the lack of knowledge or absence of information regarding the patient's medication history as barriers to performing MR in the ED14,15. Despite the short time, it is possible to implement strategies to promote the performance of this process in emergency units, such as developing protocols, training other members of the multidisciplinary team to conduct MR, using digital resources, and unified systems aiming to promote the exchange of patient's information during care transitions, to collect the BPMH and reduce errors1,3,5,19.
The primary purpose of MR is to prevent ME and medication-related harm19. Omission and dose errors were the leading ME identified after MR14. Studies from different countries comparing usual care MR, including physicians and nurses, versus pharmacist-led MR also demonstrate omission as one of the primaries ME detected and others as wrong dose, frequency, and unnecessary therapy4,6,19–21. Furthermore, evidence suggests that this process reduces the number of drug discrepancies and ME with the potential to cause patient harm as the pharmacists have the knowledge regarding medications and their characteristics4,5,20,21. However, the impact of MR on preventing possible ADR is uncertain due to the quality of the available evidence22.
The studies included in this review pointed to the short period of hospitalization and the lack of knowledge or absence of information regarding the patient's medication history as barriers to performing MR in the ED14,15. Despite the short time, it is possible to implement strategies to promote the performance of this process in emergency units, such as developing protocols, training other members of the multidisciplinary team to conduct MR, using digital resources, and unified systems aiming to promote the exchange of patient's information during care transitions, to collect the BPMH and reduce errors1,3,5,19.
Two studies of this review described other clinical pharmacists’ interventions more frequently than MR13,15. These professionals have a range of responsibilities in the emergency setting, working alongside physicians and nurses aiming to optimize pharmacotherapy, improve the safe and rational use of medications, and patient education, which is essential to increase efficiency and cost-effectiveness of care23.
MR is one of the defined patient safety goals of The Joint Commission organization, but the challenges involving this process are recognized, starting from obtaining the patients’ medication list24. The systematic and standardized process for obtaining the BPMH was not reported in all the studies analyzed. Obtaining the BPMH in the emergency setting at the appropriate time and manner is essential to determine the diagnosis and an important strategy to reduce ME and risk for patient harm4,5,19.
None of the evidence retrieved studied the outcomes related to the pharmacist-MR process. Systematic reviews have demonstrated that the pharmacist involvement in this process in the ED is economically worthwhile and is related to better outcomes as patient education to their diseases and medications, drug adherence and a reduced number of ED visits after hospital discharge1,25–27.
This study has limitations. It should be considered that there is a risk of losing existing articles in emergency hospitals due to the non-standardization of terminologies and descriptors, which makes the search for studies related to the theme difficult. Also, it is possible to evaluate that there are few national publications on the subject. Besides, in most identified studies, MR was not the primary objective. In addition, the methodology applied does not allow qualitative analysis of the studies.
This scoping review reveals the lack of evidence about the pharmacist-led MR process in the emergency setting in Brazil. The results obtained could help improve the acting of clinical pharmacists in hospital emergencies services as a strategy for patient safety, considering the importance of the presence of these professionals by national and international institutions.
FundingNo funding.
Conflict of interestsNo conflict of interests.
| Year | Author(s) | Title | Exclusion criteria (justification) |
|---|---|---|---|
| 2012 | Carvalho IV, et al. | Drug- related problems cause many admissions to a Brazilian hospital paediatric emergency unit: A prospective and observational study | Methods (congress abstract) |
| 2014 | Marques LFG, Romano-Lieber NS | Patient safety strategies for safe use of medications after hospital discharge | Methods (bibliographical research) |
| 2014 | Frizon F, et al. | Reconciliação de medicamentos em hospital universitário | Population (not conducted in the emergency setting) |
| 2014 | Costa JM, et al. | Análise de um programa para otimização da transição do cuidado de pacientes em terapia antimicrobiana | Concept (RM was not performed) |
| 2015 | Selcuk A, et al. | The potential role of clinical pharmacists in elderly patients during admission | Context (not conducted in Brazil) |
| 2015 | Kuhmmer R, et al. | Effectiveness of pharmaceutical care at discharge in the emergency department: study protocol of a randomized controlled trial | Concept (RM was not performed) |
| 2016 | Figueiredo TP de, et al. | Análise das intervençães realizadas na alta hospitalar de idosos acompanhados em serviço de orientação e referenciamento farmacotêrapeutico | Concept (RM was not performed) |
| 2016 | Mendes AE, et al. | Medication reconciliation at patient admission: A randomized controlled trial | Population (not conducted in the emergency setting) |
| 2016 | González FJ, Rousseau M | Conciliación farmacoterapéutica en las áreas de transición en la atención del paciente en un hospital pediátrico de alta complejidad | Context (not conducted in Brazil) |
| 2018 | Bonetti AF, et al. | Pharmacist-led discharge medication counselling: A scoping review | Population (Brazilian studies included were not in the emergency setting) |
| 2020 | Fernandes BD, et al. | Pharmacist-led medication reconciliation at patient discharge: A scoping review | Context (did not include Brazilian studies) |
| 2020 | Bonetti AF, et al. | Impact of pharmacist-led discharge counseling on hospital readmission and emergency department visits: A systematic review and meta-analysis | Population (Brazilian studies included were not in the emergency setting) |
| 2020 | Linden LV der, et al. | Clinical pharmacy services in older inpatients: An evidence-based review | Context (did not include Brazilian studies) |
| 2021 | Carvalho IV, et al. | Adverse drug event–related admissions to a pediatric emergency unit | Concept (RM was not performed) |
| 2021 | Gheno J, Weis AH | Care transition in hospital discharge for adult patients: integrative literature review | Concept (RM was not studied) |
| 2021 | Soares S, Trevisol DJ, Schuelter-Trevisol F. | Medication discrepancies in a hospital Southern Brazil: the importance of medication reconciliation for patient safety | Population (not conducted in the emergency setting) |
| 2021 | Alcântara T dos S, et al. | Prevalence of medication discrepancies on pediatric patients transferred between hospital wards | Concept (RM was not performed) |
| 2021 | Kelly WN, et al. | Association of pharmacist counseling with adherence, 30-day readmission, and mortality: A systematic review and meta-analysis of randomized trials | Concept (RM was not performed) |
| 2021 | Ramos JR, et al. | The impact of pharmaceutical care in multidisciplinary teams on health outcomes: Systematic review and meta-analysis | Context (did not include Brazilian studies) |
MR: medication reconciliation.
Source: The authors.
| Scoping review details | |||
| Title | Pharmacist-led medication reconciliation in emergency hospital services in Brazil: a scoping review | ||
| Objectives | To map the available research evidence related to pharmacist-led MR in emergency hospital services in Brazil, in which step of care transfer MR was conducted and its outcomes | ||
| Question | What is the available research evidence of pharmacist-led MR in emergency hospital services in Brazil? | ||
| Inclusion criteria | |||
| Population | Patients admitted to Brazilian's emergency hospitals services, independently of their gender, age, comorbidities o/and reason for admission | ||
| Concept | Pharmacist-led MR conducted on any step of care transition (admission, transfer or discharge) in an emergency hospital service | ||
| Context | Studies conducted in Brazilian's services, in private or public settings | ||
| Types of evidence source | Observational and interventional studies | ||
| Evidence source details and characteristics | |||
| Citation details | Miranda TMM, et al. Interventions performed by the clinical pharmacist in the emergency department. Einstein (São Paulo), [S.L.], v. 10, n. 1, p. 74-78, mar. 2012 | Moraes GG, et al. Atuação do farmacêutico residente em uma unidade de pronto atendimento: contribuindo para a promoção da saúde. Revista de Epidemiologia e Controle de Infecção, [S.L.], v. 6, n. 4, p. 1-4, 4 out. 2016 | Spalla LR, Castilho SR de. Medication reconciliation as a strategy for preventing medication errors. Brazilian Journal of Pharmaceutical Sciences, [S.L.], v. 52, n. 1, p. 143-150, mar. 2016. |
| Country | Brazil | Brazil | Brazil |
| Context | ED of and Teaching Hospital | ED of and Teaching Hospital | ED and dermatology, endocrinology, nephrology, pneumology, gastroenterology, neurology, cardiology wards of a Teaching Hospital |
| Participants | Adults and children | Adults | Adults |
| Pharmaceutical intervention at the ED | Pharmaceutical intervention at the ED | Pharmacist-led MR conducted at hospital admission | |
| Pharmaceutical evaluation of medical prescriptions | Pharmaceutical evaluation of medical prescriptions | MR conducted at the ED | |
| Results extracted | Pharmacist-led MR within 48-hour admission | MR as one of the pharmaceutical interventions | Mean difference of the number of medications prescribed for patients before and after admission |
| ME identified by the MR process and its classification | |||
| Frequency of acceptance of pharmaceutical interventions by prescribing physicians | |||
| Report of drug allergy history during MR process | |||
ED: emergency department; MR: medication reconciliation.
Source: The authors.
Early Access date (06/01/2022).